A novel methoxy-decorated metal–organic framework exhibiting high acetylene and carbon dioxide storage capacities†
Xing
Duan
 *a,
Yuanjing
Cui
b,
Yu
Yang
b and
Guodong
Qian
*a,
Yuanjing
Cui
b,
Yu
Yang
b and
Guodong
Qian
 *b
*b
aCollege of Materials & Environmental Engineering, Hangzhou Dianzi University, Hangzhou, 310027, PR China. E-mail: star1987@hdu.edu.cn
bState Key Laboratory of Silicon Materials, Cyrus Tang Center for Sensor Materials and Applications, School of Materials Science and Engineering, Zhejiang University, Hangzhou, 310027, PR China. E-mail: gdqian@zju.edu.cn
First published on 28th December 2016
Abstract
A methoxy-decorated novel metal–organic framework (MOF), Cu2(DTPD) (ZJU-12, H4DTPD = 5,5′-(2,6-dimethoxynaphthalene-1,5-diyl)diisophthalic acid) with optimized pore space and open metal sites, was solvothermally synthesized and structurally characterized. The activated ZJU-12a displays a moderately high BET (Brunauer–Emmett–Teller) surface area of 2316 m2 g−1. Due to the pore size of the crystal being consistent with the molecular size and kinetic diameters of C2H2 and CO2, ZJU-12a exhibits a high C2H2 storage capacity of 244 cm3 g−1 and CO2 capture capacity of 134 cm3 g−1 at room temperature.
Introduction
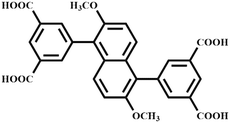
Over the past few decades, metal organic frameworks (MOFs), as a new type of crystalline porous multifunctional materials, have attracted tremendous interest from scientists in the fields of chemistry and materials science due to their exceptionally high surface area and useful and tunable porosity.1–3 Meanwhile, the pore surfaces and sizes of metal organic frameworks can be systematically functionalized and varied/tuned because of their diversity in architecture and composition, so MOF materials show rapid development in a variety of application areas, including catalysis,4 gas storage,2a,f–h molecular separation,2d drug delivery,5 and luminescence.2c Although numerous MOFs used for storage of small molecule gases have been reported and studied, quite a lot of efforts have been pursued to further enhance their gas adsorption capacity.To develop microporous metal organic framework materials with high gas adsorption performance, a series of strategies, such as improving the density of metal open sites, increasing the pore volume and surface areas, enhancing the quality of crystals and optimizing the activation conditions, taking advantage of framework catenation and interpenetration, adjusting the pore sizes and modifying the pore surfaces, has been extensively explored.6 Designing and synthesizing MOFs with open metal sites (OMSs) is one of the best strategies to enhance gas storage properties. The well-known MOF material HKUST-1 (ref. 7) with open copper coordination sites in a large cuboctahedral cage has a high acetylene adsorption capacity of 201 cm3 g−1. Xiang et al.8 reported a porous material with high densities of open metal sites, CoMOF-74, which exhibits a high volumetric acetylene storage capacity of 230 cm3 cm−3. However, high densities of open metal sites result in high adsorption enthalpy, which goes against the regeneration of crystal materials. On the other hand, pore size also plays an important role in the aspect of gas adsorption. For promoting the capability of capturing gas molecules, maximizing the guest–framework interaction by matching the pore space with the size of the gas molecules can be identified as an effective approach. To adjust the pore size, several strategies have been reported, such as inserting a symmetry-matching regulated secondary linker or anchoring a metal ion/cluster/functional groups at the cage/channel centers, which make the pore space of the primary framework partition into multiple domains.6d–j Zhao et al.6d introduced a tripyridyl-type linker into a MIL-88-type structure to obtain a family of CPM-33 materials which exhibit superior CO2 adsorption capacity. In particular, CPM-33b shows the highest CO2 adsorption value of 126.4 cm3 g−1 among MOFs without OMSs and is comparable to ZnMOF-74 and NiMOF-74.2b,9 Herein, with the purpose of improving the gas uptake capacity, we substitute benzene with dimethoxynaphthalene into terphenyl-3,3′,5,5′-tetracarboxylic acid to obtain a new tetracarboxylic organic linker H4DTPD (Scheme 1) and its corresponding first microporous MOF, [Cu2(DTPD)(H2O)2]·(DMF)5·(H2O)2 (ZJU-12; H4DTPD = 5,5′-(2,6-dimethoxynaphthalene-1,5-diyl)diisophthalic acid, ZJU = Zhejiang University), with optimized pore space and open metal sites. The activated ZJU-12a exhibits a moderately high BET (Brunauer–Emmett–Teller) surface area of 2316 m2 g−1 and a high C2H2 storage capacity of 244 cm3 g−1 and CO2 capture capacity of 134 cm3 g−1 at room temperature.
Experimental
Synthesis of the organic linker
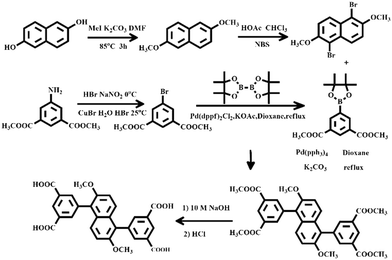
H4DTPD was synthesized as shown in Scheme 2.Dimethyl 5-amino-isophthalate (50 g) was added to 15% hydrobromic acid (900 mL) and cooled to 0 °C. A sodium nitrite solution (2.5 M, 120 mL) was added slowly with stirring to obtain a solution of diazonium bromide. The solution of diazonium bromide was slowly added to a solution including CuBr (49 g) and 15% hydrobromic acid (450 mL) under stirring, while the temperature was always kept under 0 °C. The mixture was kept stirring under room temperature overnight after the addition was completed. The solution was filtered, and the filter cake was dissolved in CCl2H2, dried with MgSO4, filtered and concentrated in a vacuum. The crude product was purified by column chromatography (silica gel, ethyl acetate/petroleum ether, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 v/v) to obtain dimethyl 5-bromobenzene-1,3-dicarboxylate as a white powder. Yield: 85%. 1H-NMR (500 MHz, CDCl3): δ = 3.95 (s, 6H), 8.35 (d, 2H), 8.61 (s, 1H) ppm.
8 v/v) to obtain dimethyl 5-bromobenzene-1,3-dicarboxylate as a white powder. Yield: 85%. 1H-NMR (500 MHz, CDCl3): δ = 3.95 (s, 6H), 8.35 (d, 2H), 8.61 (s, 1H) ppm.
The dimethyl 5-bromobenzene-1,3-dicarboxylate (5.4 g), bis(pinacolato)diborane (6.0 g), Pd(dppf)2Cl2 (0.2 g) and potassium acetate (5.6 g) were added to 100 mL of dried 1,4-dioxane. The mixture was kept at 70 °C for 24 h under stirring. Afterwards, the resultant mixture was extracted with 50 mL of ethyl acetate. The organic layer was separated and dried with anhydrous MgSO4. Then, the solvent was concentrated in a vacuum after filtration. Finally, the crude product was purified by column chromatography (silica gel, ethyl acetate/petroleum ether, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 v/v) to obtain pure dimethyl (5-pinacolboryl)isophthalate. Yield: 66%. 1H NMR (500 MHz, CDCl3): δ = 1.37 (m, 12H), 3.95 (s, 6H), 8.64 (d, 2H), 8.76 (s, 1H) ppm.
8 v/v) to obtain pure dimethyl (5-pinacolboryl)isophthalate. Yield: 66%. 1H NMR (500 MHz, CDCl3): δ = 1.37 (m, 12H), 3.95 (s, 6H), 8.64 (d, 2H), 8.76 (s, 1H) ppm.
Naphthalene-2,6-diol (10 g) and K2CO3 (20 g) were dissolved in DMF (50 mL), then iodomethane (10 mL) was added, and the mixture was kept under 85 °C for 3 hours and afterwards was filtered after adding water (100 mL). The crude product was purified via recrystallization of CCl2H2 to obtain 2,6-dimethoxynaphthalene. Yield: 85%. 1H NMR (500 MHz, CDCl3): δ = 3.90 (s, 6H), 7.66 (d, 2H), 7.12 (s, 2H), 7.15 (d, 2H) ppm.
A solution of 2,6-dimethoxynaphthalene (5 g) in a 100 mL mixed solvent of CHCl3 and acetic acid (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was added to NBS (9.6 g) under 0 °C. After the mixture reacted overnight at room temperature, the mixture was filtered. The solid was washed with water, saturated NaHCO3 solution, ethanol and CHCl3 to afford 1,5-dibromo-2,6-dimethoxynaphthalene as a white solid. Yield: 87%. 1H NMR (500 MHz, CDCl3): δ = 4.04 (s, 6H), 8.26 (d, 2H), 7.37 (d, 2H) ppm.
1) was added to NBS (9.6 g) under 0 °C. After the mixture reacted overnight at room temperature, the mixture was filtered. The solid was washed with water, saturated NaHCO3 solution, ethanol and CHCl3 to afford 1,5-dibromo-2,6-dimethoxynaphthalene as a white solid. Yield: 87%. 1H NMR (500 MHz, CDCl3): δ = 4.04 (s, 6H), 8.26 (d, 2H), 7.37 (d, 2H) ppm.
1,5-Dibromo-2,6-dimethoxynaphthalene (3 g), dimethyl (5-pinacolboryl)isophthalate (5.57 g) and K2CO3 (8 g) were added to 100 mL of anhydrous 1,4-dioxane, and the solution was deaerated under Ar for 15 min. Pd(PPh3)4 (0.47 g) was added to the reaction mixture with stirring, and the mixture was heated to 80 °C for 3 days under Ar. Afterwards, it was extracted with trichloromethane (150 mL). The organic layer was separated and was dried with anhydrous MgSO4 and the solvent was removed in a vacuum. The crude product was purified by column chromatography to obtain tetramethyl 5,5′-(2,6-dimethoxynaphthalene-1,5-diyl)diisophthalate. Yield: 59.2%. 1H-NMR (500 MHz, CDCl3): δ = 3.77 (s, 6H), 3.96 (s, 12H), 8.27 (s, 4H), 8.77 (s, 2H), 7.24 (d, 2H), 7.43 (d, 2H) ppm.
Tetramethyl 5,5′-(2,6-dimethoxynaphthalene-1,5-diyl)diisophthalate (5 g) was then suspended in 150 mL of NaOH (13.9 g) aqueous solution, and 1,4-dioxane (40 mL) was added. The mixture was stirred under reflux until clarification. Dilute HCl was added to the aqueous solution until the pH of the solution was 2. The solid was collected by filtration, washed with a lot of water, and dried to give 5,5′-(2,6-dimethoxynaphthalene-1,5-diyl)diisophthalic acid (H4DTPD, 96.7% yield). 1H-NMR (500 MHz, DMSO): δ = 3.76 (s, 6H), 7.38 (d, 2H), 7.50 (d, 2H), 8.09 (s, 4H), 8.56 (s, 2H), 13.39 (s, 4H) ppm.
Synthesis of ZJU-12
In a 20 ml glass vial, the organic linker H4DTPD (10 mg, 0.0194 mmol) was dissolved in 12.85 mL of N,N-dimethyl formamide (DMF). Then, Cu(NO3)2·2.5H2O (20 mg, 0.086 mmol) and 2.15 mL of H2O were added into the vial, followed by 75 μL of HCl (37%, aq.). The glass vial was capped and placed in a precision oven at 80 °C for 3 days. Green rhombic shaped crystals were obtained after cooling to room temperature and washed with DMF three times to afford ZJU-12. Elemental analysis: calcd for [Cu2(C28H16O10)(H2O)2]·(DMF)5·(H2O)2 (C43H59Cu2N5O19, %): C, 48.04; H, 5.36; N, 6.52; found: C, 48.075; H, 5.564; N, 6.707.Gas sorption measurements
A Micromeritics ASAP 2020 surface area analyzer was used to measure gas sorption isotherms. To obtain a guest-free framework, the fresh as-synthesized sample of ZJU-12 was washed with DMF, guest-exchanged with dry acetone at least 10 times, filtered and vacuumed at room temperature for 24 h and then at 383 K until the outgas rate was 5 μmHg min−1 prior to measurements. An activated sample of ZJU-12 (100–120 mg) was used for the sorption measurements. The N2 sorption measurement was maintained at 77 K with liquid nitrogen. The C2H2 and CO2 sorption measurements were performed at 273 K with an ice-water bath and at 298 K with a water bath.Results and discussion
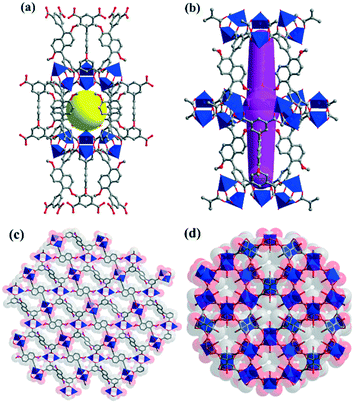
The organic ligand H4DTPD was successfully synthesized by addition reaction and Suzuki cross-coupling followed by hydrolysis of the ester and acidification. ZJU-12 was obtained by the solvothermal reaction of H4DTPD and Cu(NO3)2·2.5H2O in DMF/H2O with addition of a small quantity of HCl at 80 °C for 3 days as blue rhombic shaped crystals. The formulae of ZJU-12 were confirmed by single-crystal X-ray diffraction studies, elemental analyses and thermogravimetric analysis (TGA, Fig. S1†). The consistency of the powder X-ray diffraction (PXRD) pattern of the as-synthesized samples with the simulated pattern from single-crystal data demonstrates the phase purity of the bulk crystal materials (Fig. S2†).The single crystal X-ray crystallography (SXRD) analysis proved that ZJU-12 crystallizes in the trigonal space group R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m. As expected, ZJU-12 is built from paddle-wheel Cu2(COO)4 secondary building units (SBUs) connected to H4DTPD linkers via carboxylate groups to have the well-known NbO topology (Fig. 1). The 3D framework of ZJU-12 has two types of cages along the c axis. The diameter of the spherical-like cage is about 6.4 Å, taking into account the van der Waals radii (Fig. 1a). Furthermore, the size of the irregular shuttle-shaped cage is approximately 3.2 × 20.6 Å2 (Fig. 1b). The kinetic diameter and molecular dimensional size of C2H2 (3.3 Å, 3.32 × 3.34 × 5.7 Å3) and CO2 (3.3 Å, 3.18 × 3.33 × 5.36 Å3) are a little lower than those of the cages, therefore these two gases can easily enter the cages of the crystal. Furthermore, due to the introduction of dimethoxynaphthalene, there are smaller sizes of windows along the a, b and c axes in ZJU-12 materials compared with the prototype framework NOTT-101. One, which has a value of about 1 Å, can be observed along the c axis (Fig. 1c) and the other two along the a axis are about 2 Å and 2.8 × 5.6 Å2 (Fig. 1d). The smaller window sizes can effectively prevent gas molecules from escaping from the pore space of framework materials. The calculated accessible free pore volume of ZJU-12a is 62% (7414 Å3 out of 11
m. As expected, ZJU-12 is built from paddle-wheel Cu2(COO)4 secondary building units (SBUs) connected to H4DTPD linkers via carboxylate groups to have the well-known NbO topology (Fig. 1). The 3D framework of ZJU-12 has two types of cages along the c axis. The diameter of the spherical-like cage is about 6.4 Å, taking into account the van der Waals radii (Fig. 1a). Furthermore, the size of the irregular shuttle-shaped cage is approximately 3.2 × 20.6 Å2 (Fig. 1b). The kinetic diameter and molecular dimensional size of C2H2 (3.3 Å, 3.32 × 3.34 × 5.7 Å3) and CO2 (3.3 Å, 3.18 × 3.33 × 5.36 Å3) are a little lower than those of the cages, therefore these two gases can easily enter the cages of the crystal. Furthermore, due to the introduction of dimethoxynaphthalene, there are smaller sizes of windows along the a, b and c axes in ZJU-12 materials compared with the prototype framework NOTT-101. One, which has a value of about 1 Å, can be observed along the c axis (Fig. 1c) and the other two along the a axis are about 2 Å and 2.8 × 5.6 Å2 (Fig. 1d). The smaller window sizes can effectively prevent gas molecules from escaping from the pore space of framework materials. The calculated accessible free pore volume of ZJU-12a is 62% (7414 Å3 out of 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 960.5 Å3), evaluated by the PLATON program.10
960.5 Å3), evaluated by the PLATON program.10
To estimate the permanent porosity, the dry acetone-exchanged ZJU-12 was activated under high vacuum to obtain the desolvated ZJU-12a. The N2 adsorption isotherm of ZJU-12a, which exhibits reversible type-I adsorption behaviour, was acquired at 77 K. The highest adsorbed amount of N2 for ZJU-12a is 606 cm3 g−1 and the corresponding pore volume is 0.938 cm3 g−1 (Fig. 2). The surface areas of ZJU-12a are evaluated to be 2316 m2 g−1 from Brunauer–Emmett–Teller (BET) (Fig. S3†) and 2567 m2 g−1 from Langmuir surface areas. The BET value is slightly lower than that for NOTT-101 (ref. 11a) (2805 m2 g−1) because of substitution of benzene with dimethoxynaphthalene, but is comparable to those for ZJU-25a11b (2124 m2 g−1), NOTT-109 (ref. 11a) (2110 m2 g−1), NJU-Bai-14 (ref. 11c) (2384 m2 g−1), ZJNU-54a11d (2134 m2 g−1) and ZJNU-44 (ref. 11e) (2314 m2 g−1). The pore size distribution calculated by the Horvath–Kawazoe model is in the range from 4 Å to 6 Å, which is consistent with the pore sizes from SXRD (Fig. S4†).
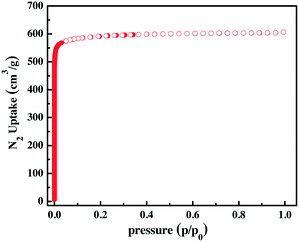 | ||
| Fig. 2 N2 sorption isotherm of ZJU-12a at 77 K. Solid and open symbols represent adsorption and desorption, respectively. | ||
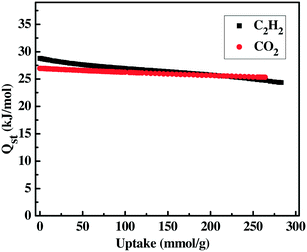
Acetylene is one of the important raw materials in industrial production, but the storage of acetylene is required to be under suitable low pressure, otherwise it has the possibility of explosion. Among all kinds of porous materials, MOF materials are considered to be the most promising ones for such an application. The optimized pore space and open metal sites within the framework of ZJU-12a encourage us to study its adsorption performance for acetylene and methane. We examine the acetylene adsorption of ZJU-12a. As shown in Fig. 3a, ZJU-12a can take up a large amount of acetylene. At 273 K and 298 K, the acetylene saturated adsorption capacity of ZJU-12a can reach 301 cm3 g−1 and 244 cm3 g−1, respectively. The gravimetric acetylene storage capacity of ZJU-12a at room temperature is significantly higher than those of examined porous MOF materials.2f,7,12 In fact, the gravimetric C2H2 adsorption capacity of 244 cm3 g−1 is the highest one ever reported (Table 1), indicating the promise of this novel microporous MOF material for practical acetylene storage applications. Taking the framework density of 0.799 g cm−3 without the coordinated waters into account, the volumetric capacities are 241 cm3 cm−3 and 195 cm3 cm−3 at 273 K and 298 K, respectively. The initial Qst of C2H2 adsorption in ZJU-12a is calculated to be 29 kJ mol−1 (Fig. 4), which is significantly lower than that for MOF-74 with high densities of open metal sites.
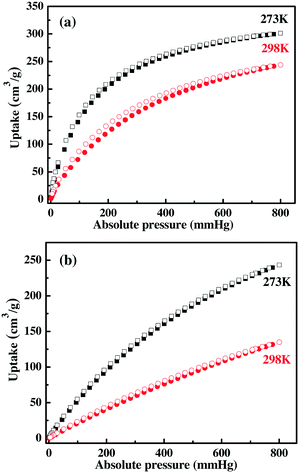 | ||
| Fig. 3 Gas sorption isotherms of ZJU-12a for (a) C2H2 at 273 K and 298 K, and (b) CO2 at 273 K and 298 K. Solid symbols: adsorption, open symbols: desorption. | ||
| MOFs | S BET (m2 g−1) | V P (cm3 g−1) | C2H2 uptake (cm3 g−1) | Ref. |
|---|---|---|---|---|
| ZJU-12a | 2316 | 0.938 | 244 | This work |
| FJI-H8 | 2025 | 0.82 | 224 | 12e |
| NJU-Bai17 | 2423 | 0.914 | 222.4 | 12g |
| ZJU-40a | 2858 | 1.06 | 216 | 12o |
| ZJNU-47 | 2638 | 1.031 | 213 | 12h |
| ZJNU-54 | 2134 | 0.871 | 211 | 11d |
| Cu 2 TPTC-OMe | 2278 | 1.039 | 204 | 12j |
| Cu 2 TPTC-Me | 2405 | 0.9805 | 203 | 12j |
| HKUST-1 | 1850 | 0.76 | 201 | 7 |
| CoMOF-74 | 1504 | 0.63 | 197 | 12b |
| ZJU-8a | 2501 | 1.0224 | 195 | 12k |
| ZJU-5a | 2823 | 1.074 | 193 | 12f |
| ZJU-9a | 2353 | 0.887 | 193 | 12l |
| ZJU-70 | 1362 | 0.676 | 191 | 12c |
| NOTT-101 | 2805 | 1.080 | 184 | 12d |
| ZJU-7a | 2198 | 0.8945 | 180 | 12m |
| PCN-16 | 2810 | 1.06 | 176 | 12a |
| MOF-505 | 1139 | 0.67 | 148 | 7 |
| NOTT-102 | 3342 | 1.280 | 146 | 12d |
| ZJU-26a | 989 | 0.572 | 84 | 12n |
The excessive emission of carbon dioxide (CO2) is the major factor for the greenhouse effect. MOF materials are very promising solid porous adsorbents for CO2 capture on account of their intrinsic advantages. We investigate the CO2 adsorption performance, indicating that ZJU-12a exhibits a CO2 uptake of 243 cm3 g−1 at 273 K and 134 cm3 g−1 at 298 K at 1 bar (Fig. 3b). Remarkably, the gravimetric CO2 sorption capacity at room temperature is higher than those of MOF materials ZJNU-54a12i (120 cm3 g−1), PCN-124 (ref. 13a) (114 cm3 g−1), ZJNU-44 (ref. 13b) (116 cm3 g−1), NPC-6 (ref. 13c) (108 cm3 g−1), JLU-Liu21 (ref. 13d) (118 cm3 g−1), NJU-Bai21 (ref. 13e) (115 cm3 g−1) and NJU-Bai-14 (ref. 11c) (100 cm3 g−1). The high C2H2 and CO2 adsorption values demonstrate that open metal sites and optimized pore size can really enhance the interaction between the gas molecules and the framework. The initial adsorption enthalpy Qst for CO2 was calculated by the virial method and the value is 26.9 kJ mol−1 (Fig. 4). Such low C2H2 and CO2 adsorption enthalpy values meet the requirement of low energy consumption in the adsorbent recycling process.
Conclusions
In summary, by substituting benzene with dimethoxynaphthalene within the framework, we have developed a three-dimensional microporous metal–organic framework, ZJU-12. The activated ZJU-12a exhibits a moderately high BET surface area of 2316 m2 g−1. The optimized pore space and open metal sites within ZJU-12a have enabled this novel framework material to adsorb a large amount of C2H2 (244 cm3 g−1) and CO2 (134 cm3 g−1) at room temperature. Thus, this new MOF can be a promising adsorbent for gas storage applications.Acknowledgements
The authors gratefully acknowledge financial support from the National Natural Science Foundation of China (No. 51472217, 51432001 and 51602087) and the Zhejiang Provincial Natural Science Foundation of China (No. LZ15E020001).Notes and references
-
(a) M. Eddaoudi, J. Kim, N. Rosi, D. Vodak, J. Wachter, M. O'Keeffe and O. M. Yaghi, Science, 2002, 295, 469–472 CrossRef CAS PubMed
; (b) M. P. Suh, H. J. Park, T. K. Prasad and D.-W. Lim, Chem. Rev., 2012, 112, 782–835 CrossRef CAS PubMed
; (c) H. Furukawa, N. Ko, Y. B. Go, N. Aratani, S. B. Choi, E. Choi, A. Ö. Yazaydin, R. Q. Snurr, M. O'Keeffe, J. Kim and O. M. Yaghi, Science, 2010, 329, 424–428 CrossRef CAS PubMed
; (d) E. D. Bloch, W. L. Queen, R. Krishna, J. M. Zadrozny, C. M. Brown and J. R. Long, Science, 2012, 335, 1606–1610 CrossRef CAS PubMed
; (e) J.-R. Li, J. Sculley and H.-C. Zhou, Chem. Rev., 2012, 112, 869–932 CrossRef CAS PubMed
; (f) S. S.-Y. Chui, S. M.-F. Lo, J. P. H. Charmant, A. G. Orpen and I. D. Williams, Science, 1999, 283, 1148–1150 CrossRef CAS PubMed
; (g) B. Chen, M. Eddaoudi, S. T. Hyde, M. O'Keeffe and O. M. Yaghi, Science, 2001, 291, 1021–1023 CrossRef CAS PubMed
.
-
(a) Y. He, W. Zhou, G. Qian and B. Chen, Chem. Soc. Rev., 2014, 43, 5657–5678 RSC
; (b) K. Sumida, D. L. Rogow, J. A. Mason, T. M. McDonald, E. D. Bloch, Z. R. Herm, T.-H. Bae and J. R. Long, Chem. Rev., 2012, 112, 724–781 CrossRef CAS PubMed
; (c) Y. Cui, Y. Yue, G. Qian and B. Chen, Chem. Rev., 2012, 112, 1126–1162 CrossRef CAS PubMed
; (d) B. Li, H. Wang and B. Chen, Chem. – Asian J., 2014, 9, 1474–1498 CrossRef CAS PubMed
; (e) H.-L. Jiang, T. A. Makal and H.-C. Zhou, Coord. Chem. Rev., 2013, 257, 2232–2249 CrossRef CAS
; (f) Z. Zhang, S. Xiang and B. Chen, CrystEngComm, 2011, 13, 5983–5992 RSC
; (g) Z. Zhang, Z.-Z. Yao, S. Xiang and B. Chen, Energy Environ. Sci., 2014, 7, 2868–2899 RSC
; (h) Y. He, W. Zhou, R. Krishna and B. Chen, Chem. Commun., 2012, 48, 11813–11831 RSC
; (i) Y. Bai, Y. Dou, L.-H. Xie, W. Rutledge, J.-R. Li and H.-C. Zhou, Chem. Soc. Rev., 2016, 45, 2327–2367 RSC
; (j) W. Lu, Z. Wei, Z. Gu, T. Liu, J. Park, J. Park, J. Tian, M. Zhang, Q. Zhang, T. Gentle III, M. Bosch and H. C. Zhou, Chem. Soc. Rev., 2014, 43, 5561–5593 RSC
.
-
(a) T. M. McDonald, J. A. Mason, X. Kong, E. D. Bloch, D. Gygi, A. Dani, V. Crocella, F. Giordanino, S. O. Odoh, W. S. Drisdell, B. Vlaisavljevich, A. L. Dzubak, R. Poloni, S. K. Schnell, N. Planas, K. Lee, T. Pascal, L. F. Wan, D. Prendergast, J.
B. Neaton, B. Smit, J. B. Kortright, L. Gagliardi, S. Bordiga, J. A. Reimer and J. R. Long, Nature, 2015, 519, 303–308 CrossRef CAS PubMed
; (b) K. Koh, A. G. Wong-Foy and A. J. Matzger, J. Am. Chem. Soc., 2009, 131, 4184–4185 CrossRef CAS PubMed
; (c) Y.-X. Tan, Y.-P. He and J. Zhang, RSC Adv., 2015, 5, 7794–7797 RSC
; (d) S. Yang, X. Lin, W. Lewis, M. Suyetin, E. Bichoutskaia, J. E. Parker, C. C. Tang, D. R. Allan, P. J. Rizkallah, P. Hubberstey, N. R. Champness, K. Mark Thomas, A. J. Blake and M. Schröder, Nat. Mater., 2012, 11, 710–716 CrossRef CAS PubMed
; (e) H. Wu, W. Zhou and T. Yildirim, J. Am. Chem. Soc., 2009, 131, 4995–5000 CrossRef CAS PubMed
; (f) T.-L. Hu, H. Wang, B. Li, R. Krishna, H. Wu, W. Zhou, Y. Zhao, Y. Han, X. Wang, W. Zhu, Z. Yao, S. Xiang and B. Chen, Nat. Commun., 2015, 6, 7328–7336 CrossRef CAS PubMed
; (g) J. Jiang, H. Furukawa, Y.-B. Zhang and O. M. Yaghi, J. Am. Chem. Soc., 2016, 138, 10244–10251 CrossRef CAS PubMed
; (h) Y. Cui, B. Li, H. He, W. Zhou, B. Chen and G. Qian, Acc. Chem. Res., 2016, 49, 483–493 CrossRef CAS PubMed
.
-
(a) T. Zhang, K. Manna and W. Lin, J. Am. Chem. Soc., 2016, 138, 3241–3249 CrossRef CAS PubMed
; (b) M. Zhao, S. Ou and C.-D. Wu, Acc. Chem. Res., 2014, 47, 1199–1207 CrossRef CAS PubMed
; (c) A. Corma, H. Garcia and F. X. Llabrés i Xamena, Chem. Rev., 2010, 110, 4606–4655 CrossRef CAS PubMed
; (d) A. H. Chughtai, N. Ahmad, H. A. Younus, A. Laypkov and F. Verpoort, Chem. Soc. Rev., 2015, 44, 6804–6849 RSC
.
-
(a) A. C. McKinlay, R. E. Morris, P. Horcajada, G. Férey, R. Gref, P. Couvreur and C. Serre, Angew. Chem., Int. Ed., 2010, 49, 6260–6266 (
Angew. Chem.
, 2010
, 122
, 6400–6406
) CrossRef CAS PubMed
; (b) J. Della Rocca, D. Liu and W. Lin, Acc. Chem. Res., 2011, 44, 957–968 CrossRef CAS PubMed
; (c) P. Horcajada, R. Gref, T. Baati, G. Maurin, P. Couvreur, G. Ferey, R. E. Morris and C. Serre, Chem. Rev., 2012, 112, 1232–1268 CrossRef CAS PubMed
; (d) C.-Y. Sun, C. Qin, X.-L. Wang and Z.-M. Su, Expert Opin. Drug Delivery, 2013, 10, 89–101 CrossRef CAS PubMed
.
-
(a) H. K. Chae, D. Y. Siberio-Perez, J. Kim, Y. Go, M. Eddaoudi, A. J. Matzger, M. O'Keeffe and O. M. Yaghi, Nature, 2004, 427, 523–527 CrossRef CAS PubMed
; (b) B. Kesanli, Y. Cui, M. R. Smith, E. W. Bittner, B. C. Bockrath and W. Lin, Angew. Chem., Int. Ed., 2005, 44, 72–75 CrossRef CAS PubMed
; (c) S. S. Kaye, A. Dailly, O. M. Yaghi and J. R. Long, J. Am. Chem. Soc., 2007, 129, 14176–14177 CrossRef CAS PubMed
; (d) X. Zhao, X. Bu, Q.-G. Zhai, H. Tran and P. Feng, J. Am. Chem. Soc., 2015, 137, 1396–1399 CrossRef CAS PubMed
; (e) S.-T. Zheng, J. T. Bu, Y. Li, T. Wu, F. Zuo, P. Feng and X. Bu, J. Am. Chem. Soc., 2010, 132, 17062–17064 CrossRef CAS PubMed
; (f) S.-T. Zheng, C. Mao, T. Wu, S. Lee, P. Feng and X. Bu, J. Am. Chem. Soc., 2012, 134, 11936–11939 CrossRef CAS PubMed
; (g) S.-T. Zheng, T. Wu, F. Zuo, C.-T. Chou, P. Feng and X. Bu, J. Am. Chem. Soc., 2012, 134, 1934–1937 CrossRef CAS PubMed
; (h) F. Bu, Q. Lin, Q. Zhai, L. Wang, T. Wu, S.-T. Zheng, X. Bu and P. Feng, Angew. Chem., Int. Ed., 2012, 51, 8538–8541 CrossRef CAS PubMed
; (i) S.-T. Zheng, T. Wu, B. Irfanoglu, F. Zuo, P. Feng and X. Bu, Angew. Chem., Int. Ed., 2011, 50, 8034–8037 CrossRef CAS PubMed
; (j) S.-T. Zheng, X. Zhao, S. Lau, A. Fuhr, P. Feng and X. Bu, J. Am. Chem. Soc., 2013, 135, 10270–10273 CrossRef CAS PubMed
.
- S. Xiang, W. Zhou, J. M. Gallegos, Y. Liu and B. Chen, J. Am. Chem. Soc., 2009, 131, 12415–12419 CrossRef CAS PubMed
.
- S. Xiang, Y. He, Z. Zhang, H. Wu, W. Zhou, R. Krishna and B. Chen, Nat. Commun., 2012, 3, 954–962 CrossRef PubMed
.
- S. R. Caskey, A. G. Wong-Foy and A. J. Matzger, J. Am. Chem. Soc., 2008, 130, 10870–10871 CrossRef CAS PubMed
.
-
L. Spek, PLATON, The University of Utrecht, Utrecht, The Netherlands, 1999 Search PubMed
.
-
(a) Y. He, W. Zhou, T. Yildirim and B. Chen, Energy Environ. Sci., 2013, 6, 2735–2744 RSC
; (b) X. Duan, J. Yu, J. Cai, Y. He, C. Wu, W. Zhou, T. Yildirim, Z. Zhang, S. Xiang, M. O'Keeffe, B. Chen and G. Qian, Chem. Commun., 2013, 49, 2043–2045 RSC
; (c) M. Zhang, Q. Wang, Z. Lu, H. Liu, W. Liu and J. Bai, CrystEngComm, 2014, 16, 6287–6290 RSC
; (d) J. Jiao, L. Dou, H. Liu, F. Chen, D. Bai, Y. Feng, S. Xiong, D.-L. Chen and Y. He, Dalton Trans., 2016, 45, 13373–13382 RSC
; (e) C. Song, J. Hu, Y. Ling, Y.-L. Feng, R. Krishna, D.-L. Chen and Y. He, J. Mater. Chem. A, 2015, 3, 19417–19426 RSC
.
-
(a) Y. Hu, S. Xiang, W. Zhang, Z. Zhang, L. Wang, J. Bai and B. Chen, Chem. Commun., 2009, 7551–7553 RSC
; (b) S. Xiang, W. Zhou, Z. Zhang, M. A. Green, Y. Liu and B. Chen, Angew. Chem., Int. Ed., 2010, 49, 4615–4618 CAS
; (c) X. Duan, C. Wu, S. Xiang, W. Zhou, T. Yildirim, Y. Cui, Y. Yang, B. Chen and G. Qian, Inorg. Chem., 2015, 54, 4377–4381 CrossRef CAS PubMed
; (d) Y. He, R. Krishna and B. Chen, Energy Environ. Sci., 2012, 5, 9107–9120 RSC
; (e) J. Pang, F. Jiang, M. Wu, C. Liu, K. Su, W. Lu, D. Yuan and M. Hong, Nat. Commun., 2015, 6, 7575–7581 CrossRef PubMed
; (f) X. Rao, J. Cai, J. Yu, Y. He, C. Wu, W. Zhou, T. Yildirim, B. Chen and G. Qian, Chem. Commun., 2013, 49, 6719–6721 RSC
; (g) M. Zhang, B. Li, Y. Li, Q. Wang, W. Zhang, B. Chen, S. Li, Y. Pan, X. You and J. Bai, Chem. Commun., 2016, 52, 7241–7244 RSC
; (h) C. Song, J. Jiao, Q. Lin, H. Liu and Y. He, Dalton Trans., 2016, 45, 4563–4569 RSC
; (i) J. Jiao, L. Dou, H. Liu, F. Chen, D. Bai, Y. Feng, S. Xiong, D.-L. Chen and Y. He, Dalton Trans., 2016, 45, 13373–13382 RSC
; (j) T. Xia, J. Cai, H. Wang, X. Duan, Y. Cui, Y. Yang and G. Qian, Microporous Mesoporous Mater., 2015, 215, 109–115 CrossRef CAS
; (k) J. Cai, H. Wang, H. Wang, X. Duan, Z. Wang, Y. Cui, Y. Yang, B. Chen and G. Qian, RSC Adv., 2015, 5, 77417–77422 RSC
; (l) X. Duan, H. Wang, Y. Cui, Y. Yang, Z. Wang, B. Chen and G. Qian, RSC Adv., 2015, 5, 84446–84450 RSC
; (m) J. Cai, Y. Lin, J. Yu, C. Wu, L. Chen, Y. Cui, Y. Yang, B. Chen and G. Qian, RSC Adv., 2014, 4, 49457–49461 RSC
; (n) X. Duan, J. Cai, J. Yu, C. Wu, Y. Cui, Y. Yang and G. Qian, Microporous Mesoporous Mater., 2013, 181, 99–104 CrossRef CAS
; (o) H. Wen, H. Wang, B. Li, Y. Cui, H. Wang, G. Qian and B. Chen, Inorg. Chem., 2016, 55, 7214–7218 CrossRef CAS PubMed
.
-
(a) J. Park, J.-R. Li, Y.-P. Chen, J. Yu, A. A. Yakovenko, Z. U. Wang, L.-B. Sun, P. B. Balbuena and H.-C. Zhou, Chem. Commun., 2012, 48, 9995–9997 RSC
; (b) C. Song, J. Hu, Y. Ling, Y. Feng, R. Krishna, D.-L. Chen and Y. He, J. Mater. Chem. A, 2015, 3, 19417–19426 RSC
; (c) Q. Mu, H. Wang, L. Li, C. Wang, Y. Wang and X. Zhao, Chem. – Asian J., 2015, 10, 1864–1869 CrossRef CAS PubMed
; (d) B. Liu, S. Yao, C. Shi, G. Li, Q. Huo and Y. Liu, Chem. Commun., 2016, 52, 3223–3226 RSC
; (e) Z. Lu, J. Bai, C. Hang, F. Meng, W. Liu, Y. Pan and X. You, Chem. – Eur. J., 2016, 22, 6277–6285 CrossRef CAS PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 1510153. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ce02291j |
| This journal is © The Royal Society of Chemistry 2017 |