Photoreactive salt cocrystal: N+–H⋯N hydrogen bond and cation–π interactions support a cascade-like photodimerization of a 4-stilbazole†
Changan
Li
,
Gonzalo
Campillo-Alvarado
 ,
Dale C.
Swenson
and
Leonard R.
MacGillivray
,
Dale C.
Swenson
and
Leonard R.
MacGillivray
 *
*
Department of Chemistry, University of Iowa, Iowa City, IA 52242-1294, USA. E-mail: len-macgillivray@uiowa.edu
First published on 21st January 2021
Abstract
We describe hydrogen bonds (i.e., N+–H⋯N) in combination with cation⋯π interactions to enable a cascade-like [2 + 2] photodimerization of 4-stilbazole in a salt cocrystal. A four-component crystal assembly is composed of photoactive pyridinium ion pair 4-stilbzH+ (4-stilbz = trans-1-(4-pyridyl)-2-(phenyl)ethylene) and photostable molecule pair 4-stilbz. UV irradiation gives rctt-1,2-bis(4-pyridyl)-3,4-bis(phenyl)cyclobutane (4-pyr-ph-cb) in quantitative yield. An intermediate structure 2(4-stilbz)·(4-pyr-ph-cb)2H+ is isolated that undergoes a partial single-crystal-to-single-crystal transformation. The single-crystal X-ray data provides a snapshot of movements in the salt cocrystal in the course of the photodimerization.
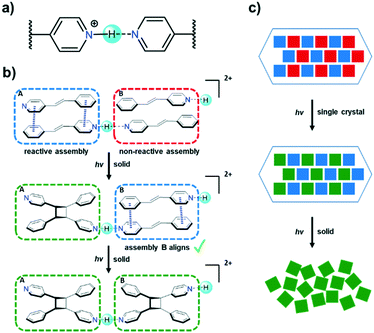
The hydrogen bond is a “master-key” interaction in crystal engineering, providing manifold opportunities to generate supramolecular materials such as polymers and photoactive solids.1 In the latter, there has in recent years been a great deal of interest to direct intermolecular [2 + 2] photodimerizations in the organic solid state using hydrogen bonds.2 Hydrogen bonds of X–H⋯X type involve a nearly linear geometry where donor and acceptor are identical. The N+–H⋯N hydrogen bond involving pyridines is, in particular, one of the most exploited X–H⋯X hydrogen bonds in crystal engineering (Scheme 1a).3
With exception to work of Peedikakkal,4 all examples involving the use of hydrogen bonds to support a photodimerization in the solid state have involved hydrogen bonds with different donor and acceptor groups (i.e., X–H⋯Y). The interaction reported here involves a highly electron-deficient pyridinium moiety and neutral pyridyl group bridged by a proton (Scheme 1a). The N⋯N distance normally lies within a distance of 2.6–2.8 Å. Importantly, X+–H⋯X type hydrogen bonds are unique from a synthetic standpoint since one of the two identical hydrogen bonding functional groups must either gain or lose a proton to support charge formation and assembly of the bridge; consequently, one of the two components is also typically charge neutral and the outcome is a salt cocrystal.
Recently, there have been reports of [2 + 2] photodimerizations that occur in the solid state to proceed as cascade-like reactions.5 In the solids, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds that do not conform to the criteria of Schmidt undergo sufficient molecular movements to facilitate photodimerization processes. As part of our efforts to investigate photoactive solids,6 herein we report use of the N+–H⋯N hydrogen bond along with the cation–π interaction to support a cascade-like [2 + 2] photodimerization of a 4-stilbz in the solid state.7 We show the combined forces to support the formation of photoactive pyridinium 4-stilbzH+ assemblies that react to give head-to-tail (ht) 4-pyr-ph-cb that involves a SCSC transformation. In the process, the photodimerization making up the SCSC reaction serves to trigger rearrangement of a photostable stacked pair of molecules (i.e., 4-stilbz) to become photoreactive. The result is the regiocontrolled formation of 4-pyr-ph-cb in quantitative yield. To our knowledge, our work represents the first case of a N+–H⋯N hydrogen bond that supports a SCSC [2 + 2] photodimerization.
C bonds that do not conform to the criteria of Schmidt undergo sufficient molecular movements to facilitate photodimerization processes. As part of our efforts to investigate photoactive solids,6 herein we report use of the N+–H⋯N hydrogen bond along with the cation–π interaction to support a cascade-like [2 + 2] photodimerization of a 4-stilbz in the solid state.7 We show the combined forces to support the formation of photoactive pyridinium 4-stilbzH+ assemblies that react to give head-to-tail (ht) 4-pyr-ph-cb that involves a SCSC transformation. In the process, the photodimerization making up the SCSC reaction serves to trigger rearrangement of a photostable stacked pair of molecules (i.e., 4-stilbz) to become photoreactive. The result is the regiocontrolled formation of 4-pyr-ph-cb in quantitative yield. To our knowledge, our work represents the first case of a N+–H⋯N hydrogen bond that supports a SCSC [2 + 2] photodimerization.
Mixing of an ethanol (1 mL) solution of ammonium hexafluorophosphate (NH4PF6, 18 mg, 0.11 mmol) with 4-stilbz (40 mg, 0.22 mmol) in tetrahydrofuran (4 mL) afforded colorless single crystals of [4-stilbz·(4-stilbzH+)(PF6−)] as irregular prisms by slow evaporation in a period of 3 days. Single-crystal X-ray diffraction and 1H NMR spectroscopy confirmed the composition of the solid as [4-stilbz·(4-stilbzH+)(PF6−)]. Individual datasets for [4-stilbz·(4-stilbzH+)(PF6−)] were collected at room temperature (298 K) and low temperature (125 K). While refinement afforded better agreement of data (i.e. lower R-value) at the lower temperature, details of the room temperature structure are mostly described here to be consistent with the conditions at which the photodimerization was conducted.
The components of [4-stilbz·(4-stilbzH+)(PF6−)] crystallize in the triclinic space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) . The asymmetric unit consists of a pyridinium ion 4-stilbzH+, 4-stilbz molecule and hexafluorophosphate (PF6−) ion that form a salt cocrystal. Each 4-stilbzH+ ion and 4-stilbz molecule are disordered over two positions and interact by N+–H⋯N hydrogen bond to form a linear two-component dimeric array (N+⋯N 2.716(17) Å; N+–H⋯N 170°) (Fig. 1a). The data at the lower temperature shows markedly less disorder and is consistent with pedal motion of the C
. The asymmetric unit consists of a pyridinium ion 4-stilbzH+, 4-stilbz molecule and hexafluorophosphate (PF6−) ion that form a salt cocrystal. Each 4-stilbzH+ ion and 4-stilbz molecule are disordered over two positions and interact by N+–H⋯N hydrogen bond to form a linear two-component dimeric array (N+⋯N 2.716(17) Å; N+–H⋯N 170°) (Fig. 1a). The data at the lower temperature shows markedly less disorder and is consistent with pedal motion of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond. The components stack in a ht orientation through face-to-face cation⋯π interactions of the pyridinium and phenyl groups. The outcome is stacking of two pyridinium ions to generate a four-component assembly (Fig. 1b). The C
C bond. The components stack in a ht orientation through face-to-face cation⋯π interactions of the pyridinium and phenyl groups. The outcome is stacking of two pyridinium ions to generate a four-component assembly (Fig. 1b). The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds of each face-to-face stacked 4-stilbzH+ assembly are parallel and separated by 3.89 Å, which falls within the criteria of Schmidt.8 Adjacent stacked assemblies are arranged offset by face-to-face molecule–molecule contacts, with the molecules (dark and light gray) stacking ht and interacting via displaced π⋯π contacts. The C
C bonds of each face-to-face stacked 4-stilbzH+ assembly are parallel and separated by 3.89 Å, which falls within the criteria of Schmidt.8 Adjacent stacked assemblies are arranged offset by face-to-face molecule–molecule contacts, with the molecules (dark and light gray) stacking ht and interacting via displaced π⋯π contacts. The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds of the stacked molecules of the salt cocrystal are separated by 4.56 Å (Fig. 1c).
C bonds of the stacked molecules of the salt cocrystal are separated by 4.56 Å (Fig. 1c).
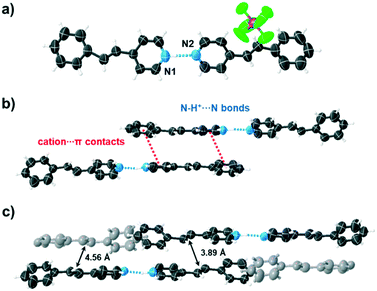 | ||
| Fig. 1 Crystal structure [4-stilbz·(4-stilbzH+)(PF6−)]: (a) two-component dimeric array, (b) four-component assembly, (c) stacking of four-component assemblies. | ||
It is only the stacked 4-stilbzH+ ions that conform to the geometry criteria for a [2 + 2] photodimerization. However, when a powdered crystalline sample of [4-stilbz·(4-stilbzH+)(PF6−)] was irradiated using UV light (450 W medium pressure Hg vapor lamp) for a period of 20 h, all C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds reacted to form 4-pyr-ph-cb in quantitative yield. The generation of the photoproduct was evidenced by the disappearance of the olefinic signal at 7.78 ppm and the appearance of the cyclobutane signal at 4.75 ppm (Fig. S1†).
C bonds reacted to form 4-pyr-ph-cb in quantitative yield. The generation of the photoproduct was evidenced by the disappearance of the olefinic signal at 7.78 ppm and the appearance of the cyclobutane signal at 4.75 ppm (Fig. S1†).
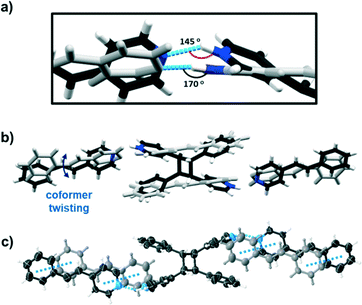
An explanation for the quantitative conversion of the stacked C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds of [4-stilbz·(4-stilbzH+)(PF6−)] can be rationalized on the basis of the solid undergoing a partial SCSC transformation. When [4-stilbz·(4-stilbzH+)(PF6−)] was subjected to UV light for a period of 8 h using a gel nail dryer as a source,9 the stacked 4-stilbzH+ ions reacted in a SCSC process to form [2(4-stilbz)·(4-pyr-ph-cb2H+)(2PF6−)] (Fig. 2). The presence of a single cyclobutane is consistent with pedal motion.10 The generation of the photoproduct was accompanied by bending of the N+–H⋯N hydrogen bond (i.e., N+–H⋯N 170° to 145°) and tilting of the 4-pyridyl moieties (Fig. 2a).11 The tilting resulted in an ca. 25 degree rotation of the 4-stilbz molecule such that the stacked molecules are engaged exclusively in face-to-face π⋯π forces (Fig. 2c). We suggest that the molecular movements enable the remaining C
C bonds of [4-stilbz·(4-stilbzH+)(PF6−)] can be rationalized on the basis of the solid undergoing a partial SCSC transformation. When [4-stilbz·(4-stilbzH+)(PF6−)] was subjected to UV light for a period of 8 h using a gel nail dryer as a source,9 the stacked 4-stilbzH+ ions reacted in a SCSC process to form [2(4-stilbz)·(4-pyr-ph-cb2H+)(2PF6−)] (Fig. 2). The presence of a single cyclobutane is consistent with pedal motion.10 The generation of the photoproduct was accompanied by bending of the N+–H⋯N hydrogen bond (i.e., N+–H⋯N 170° to 145°) and tilting of the 4-pyridyl moieties (Fig. 2a).11 The tilting resulted in an ca. 25 degree rotation of the 4-stilbz molecule such that the stacked molecules are engaged exclusively in face-to-face π⋯π forces (Fig. 2c). We suggest that the molecular movements enable the remaining C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds to react quantitatively to form 4-pyr-ph-cb, although we note the stacking distance to increase slightly from 4.56 to 4.62 Å (Fig. 2c). Accumulations and releases of stress and strain may also contribute to the C
C bonds to react quantitatively to form 4-pyr-ph-cb, although we note the stacking distance to increase slightly from 4.56 to 4.62 Å (Fig. 2c). Accumulations and releases of stress and strain may also contribute to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds reacting.12
C bonds reacting.12
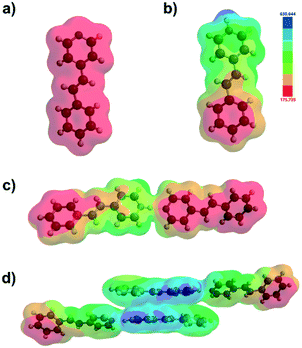
B3LYP density functional molecular calculations using 3-21G basis set in the gas phase were performed to probe the driving force of the selective face-to-face stacking of [4-stilbz·(4-stilbzH+)(PF6−)].13 Electrostatic potential maps of 4-stilbz/4-stilbzH+ monomer, the two-component dimeric array and the four-component assembly as derived from the X-ray data of [4-stilbz·(4-stilbzH+)(PF6−)] and [2(4-stilbz)·(4-pyr-ph-cb2H+)(2PF6−)] revealed the pyridinium and phenyl groups to be electron-deficient (blue) and electron-rich (red), respectively (Fig. 3). The opposite electrostatic potentials of the pyridinium and phenyl groups are complimentary and account for the face-to-face stacking of the photoactive pairs of 4-stilbzH+ ions within the salt cocrystal.
Conclusions
We have shown that the N+–H⋯N type hydrogen bond can support a [2 + 2] photodimerization in the solid state. The photodimerization proceeded in a cascade-like process via a partial SCSC transformation. We expect uses of the less common type of hydrogen bond to extend a toolbox to enable the design and synthesis of photoactive solid-state materials. The observations of the cascade-like process can also extend our understanding of dynamic processes in organic solids.Conflicts of interest
There are no conflicts to declare.Acknowledgements
We gratefully acknowledge the National Science Foundation (LRM DMR-1708673) for financial support.References
-
(a) C. B. Aakeröy and K. R. Seddon, Chem. Soc. Rev., 1993, 22, 397–407 RSC
; (b) G. R. Desiraju, Acc. Chem. Res., 2002, 35, 565–573 CrossRef CAS PubMed
; (c) B. J. Folmer, R. Sijbesma, R. Versteegen, J. Van der Rijt and E. Meijer, Adv. Mater., 2000, 12, 874–878 CrossRef CAS
; (d) L. J. Prins, D. N. Reinhoudt and P. Timmerman, Angew. Chem., Int. Ed., 2001, 40, 2382–2426 CrossRef CAS
; (e) B. Wang, R.-B. Lin, Z. Zhang, S. Xiang and B. Chen, J. Am. Chem. Soc., 2020, 142, 14399–14416 CrossRef CAS PubMed
.
-
(a) L. R. MacGillivray, J. L. Reid and J. A. Ripmeester, J. Am. Chem. Soc., 2000, 122, 7817–7818 CrossRef CAS
; (b) L. R. Macgillivray, G. S. Papaefstathiou, T. Friščić, T. D. Hamilton, D.-K. Bučar, Q. Chu, D. B. Varshney and I. G. Georgiev, Acc. Chem. Res., 2008, 41, 280–291 CrossRef CAS PubMed
; (c) S. Yamada and Y. Nojiri, Chem. Commun., 2011, 47, 9143–9145 RSC
; (d) M. Linares and A. Briceño, New J. Chem., 2010, 34, 587–590 RSC
.
-
(a) P. Gilli, V. Bertolasi, V. Ferretti and G. Gilli, J. Am. Chem. Soc., 1994, 116, 909–915 CrossRef CAS
; (b) F. A. Mautner and M. A. Goher, Polyhedron, 1998, 18, 553–559 CrossRef
; (c) R. Santra, N. Ghosh and K. Biradha, New J. Chem., 2008, 32, 1673–1676 RSC
; (d) D. Braga, S. L. Giaffreda, F. Grepioni, G. Palladino and M. Polito, New J. Chem., 2008, 32, 820–828 RSC
; (e) D. Lacková, I. Ondrejkovičová, Z. Padělková and M. Koman, J. Coord. Chem., 2014, 67, 1652–1663 CrossRef
; (f) J. Fábry, Acta Crystallogr., Sect. E: Crystallogr. Commun., 2017, 73, 1344–1347 CrossRef PubMed
.
- A. M. P. Peedikakkal, J. Chem. Sci., 2017, 129, 733–739 CrossRef CAS
.
-
(a) M. Nagarathinam and J. J. Vittal, Angew. Chem., Int. Ed., 2006, 45, 4337–4341 CrossRef CAS PubMed
; (b) S. Yamada, Y. Tokugawa, Y. Nojiri and E. Takamori, Chem. Commun., 2012, 48, 1763–1765 RSC
; (c) Z.-T. Ma, S.-Y. Yang, L.-S. Zheng and B. K. Teo, Cryst. Growth Des., 2019, 19, 3113–3119 CrossRef CAS
; (d) S. P. Yelgaonkar, D. C. Swenson and L. R. MacGillivray, Chem. Sci., 2020, 11, 3569–3573 RSC
.
-
(a) G. Campillo-Alvarado, C. Li, D. C. Swenson and L. R. MacGillivray, Cryst. Growth Des., 2019, 19, 2511–2518 CrossRef CAS
; (b) C. Li, G. Campillo-Alvarado, D. C. Swenson and L. R. MacGillivray, Inorg. Chem., 2019, 58, 12497–12500 CrossRef CAS PubMed
; (c) G. Campillo-Alvarado, K. P. D'mello, D. C. Swenson, S. V. Santhana Mariappan, H. Höpfl, H. Morales-Rojas and L. R. MacGillivray, Angew. Chem., Int. Ed., 2019, 58, 5413–5416 CrossRef CAS PubMed
.
-
(a) S. Yamada, N. Uematsu and K. Yamashita, J. Am. Chem. Soc., 2007, 129, 12100–12101 CrossRef CAS PubMed
; (b) B. Mondal, B. Captain and V. Ramamurthy, Photochem. Photobiol. Sci., 2011, 10, 891–894 CrossRef CAS PubMed
; (c) D. A. Dougherty, Acc. Chem. Res., 2013, 46, 885–893 CrossRef CAS PubMed
; (d) S. Yamada, Chem. Rev., 2018, 118, 11353–11432 CrossRef CAS PubMed
; (e) P. Li, C. Zhao, M. D. Smith and K. D. Shimizu, J. Org. Chem., 2013, 78, 5303–5313 CrossRef CAS PubMed
.
- G. Schmidt, Pure Appl. Chem., 1971, 27, 647–678 CAS
.
- T. Aung and C. A. Liberko, J. Chem. Educ., 2014, 91, 939–942 CrossRef CAS
.
-
(a) L. Kuz'mina, A. Vedernikov, S. Gromov and M. Alfimov, Crystallogr. Rep., 2019, 64, 691–712 CrossRef
; (b) A. M. P. Peedikakkal and J. J. Vittal, Chem. – Eur. J., 2008, 14, 5329–5334 CrossRef CAS PubMed
; (c) J. Quentin and L. R. MacGillivray, ChemPhysChem, 2020, 21, 154–163 CrossRef CAS PubMed
.
- M. A. Sinnwell, J. N. Blad, L. R. Thomas and L. R. MacGillivray, IUCrJ, 2018, 5, 491–496 CrossRef CAS PubMed
.
-
(a) I. Turowska-Tyrk, J. Phys. Org. Chem., 2004, 17, 837–847 CrossRef CAS
; (b) M. H. Mir, L. L. Koh, G. K. Tan and J. J. Vittal, Angew. Chem., Int. Ed., 2010, 49, 390–393 CrossRef CAS PubMed
; (c) C. Karunatilaka, D. K. Bučar, L. R. Ditzler, T. Friščić, D. C. Swenson, L. R. MacGillivray and A. V. Tivanski, Angew. Chem., Int. Ed., 2011, 50, 8642–8646 CrossRef CAS PubMed
; (d) P. e. Naumov, S. Chizhik, M. K. Panda, N. K. Nath and E. Boldyreva, Chem. Rev., 2015, 115, 12440–12490 CrossRef CAS PubMed
.
-
(a) A. A. Parent, D. H. Ess and J. A. Katzenellenbogen, J. Org. Chem., 2014, 79, 5448–5462 CrossRef CAS PubMed
; (b) Y. Zhao and D. G. Truhlar, Acc. Chem. Res., 2008, 41, 157–167 CrossRef CAS PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 2025376, 2025377 and 2052940. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/d0ce01885f |
| This journal is © The Royal Society of Chemistry 2021 |