DNA action on the growth and habit modification of NaCl crystals†
Yazhou
Qin
ac,
Dongdong
Yu
b and
Jianguang
Zhou
 *ac
*ac
aState Key Laboratory of Industrial Control Technology, Research Center for Analytical Instrumentation, College of Control Science and Engineering, Zhejiang University, 38 Zheda Road, Hangzhou 310027, PR China. E-mail: jgzhou@zju.edu.cn
bDepartment of Chemistry, Zhejiang University, Hangzhou, 310027, China
cHospital of Zhejiang University, Zhejiang University, Hangzhou, 310027, China
First published on 8th September 2017
Abstract
A large number of studies have been devoted to the preparation of different morphologies of NaCl crystals. In this work, we first use DNA as an additive to prepare hopper-like NaCl crystals and modulate the size of these hopper-like NaCl crystals using silver nitrate. This work helps us not only to understand the growth of NaCl crystals, but also to use DNA as an additive to design and synthesize different crystal structures.
In recent years, the use of additives to control the morphology of inorganic crystals has aroused the interest of many researchers due to the physical and chemical properties of crystals which depend on their composition and structure. Inorganic salts, as an important part of the crystal world, play an indispensable role both in industry and in our daily life. Numerous studies have been carried out to regulate the morphology of inorganic salts, including Na2CO3,1 Na2SO4,2–4 CaCO3,5–21 and NaCl.22–38 The growth process of crystals has a crucial role in the composition, structure and morphology of crystals. Therefore, in order to prepare crystals with specific properties, it is very important to study the mechanism of crystal growth. Recently, many research groups have been dedicated to the study of crystal growth processes. For example, García-Ruiz39 reviewed the mechanisms of the morphological changes. They reported the growth mechanisms for three minerals, including NaCl, CaCO3 and CaSO4·2H2O.
NaCl, a typical representative of inorganic salts, is indispensable both in industry and in life. It can serve as a raw material to produce caustic soda and soda ash, and also as an essential component in dairy, paper, fertilizer, textile, dyeing and pharmaceutical industries.1 However, NaCl has high hygroscopicity; a small amount of water molecules can form sodium chloride dihydrate solid bridges between the powder particles, making it unable to hold the crystal size and dispersion. In addition, the smaller the NaCl crystals, the easier the moisture absorption that could happen. Therefore, developing methods of how to engineer NaCl crystals to reduce or remove caking is pertinent. The first example of using additives to change the growth of NaCl crystals was found in 1783, by Romé de l'Isle who reported the habit change from {100} → {100} + {111} in the presence of urea.40 Since then, more and more researchers have been devoted to the study of NaCl habit modifiers. For instance, Gille41 and Spangeberg found that by evaporating the solvent, NaCl crystals could transform from {100} to {111} in the presence of formamide. The Bienfait42 group considered that the stability of {100} and {111} is related to the initial supersaturation of the solution relative to the concentration of formamide in the solution. Currently, a variety of substances are used as additives to prevent NaCl crystals from agglomerating or regulating their morphology, such as formamide,22 benzodiazepine,23 glycine,24–26 gelatin,27–29 silica gel and agarose gel,30 sodium dodecyl sulfate (SDS),31 various acids and their salts,1,32–35 polymers,25,37 and ferrocyanide.38
Compared to the above substances, DNA served as a biodegradable polyelectrolyte and can also be utilized as an inhibitor of inorganic crystal growth to form regular crystalline morphologies, which possess low immunogenicity and a clear molecular structure.43 However, studies on DNA used as an additive are rarely found. As far as we know, DNA was only used by Sommerdijk43 and Kato44 to regulate the growth process of CaCO3. Herein, we used DNA as an additive to explore its role in the growth of NaCl crystals by the solvent evaporation method. We also explored the effects of different sequences of DNA on the growth of NaCl crystals. In addition, we investigated the role of silver ions in modulating the morphology and crystallography of NaCl crystals grown in aqueous solution. As a result, a series of hopper-like NaCl crystals has been prepared on glass sheets by adding DNA into NaCl solution and the size of the hopper-like NaCl crystals was controlled by adding silver nitrate. Details of the experiments are given in the ESI.†
Four different sequences of DNA were used to prepare the NaCl crystals, which were labeled DNA-1, DNA-2, DNA-3 and DNA-4, respectively. The DNA sequences were shown as follows:
DNA-1: 5′-CCCCCCCCCCCC(T⋯⋯T)43-3′
DNA-2: 5′-CCCCCC(T⋯⋯T)50-3′
DNA-3: 5′-(A⋯⋯A)47-3′
DNA-4: 5′-(A⋯⋯A)25-3′
The NaCl crystals prepared using DNA were labeled NaCl-1, NaCl-2, NaCl-3 and NaCl-4, respectively. Fig. 1 exhibits typical optical micrographs of NaCl-1 crystals, which were prepared using 25 μl DNA-1 solution and 25 μl NaCl solution. Before mixing, the concentrations of the NaCl solution used in Fig. 1a–f were 0.15 mol L−1 (a), 0.30 mol L−1 (b), 0.75 mol L−1 (c), 1.50 mol L−1 (d), 2.25 mol L−1 (e) and 3.00 mol L−1 (f), respectively, while the concentration of the DNA-1 solution was maintained at 100 μmol L−1. It can be observed from the figure that as the NaCl solution concentration increases, the morphology of the precipitated crystals changes. When the NaCl solution concentration was 0.15 mol L−1 (Fig. 1a), the NaCl crystals formed petal-like structures, while increasing the NaCl solution concentration to 1.50 mol L−1 resulted in a very regular hopper-like morphology (Fig. 1d). When the concentration of NaCl continued to increase, the hopper-like crystals started to show defects, which we encircle in Fig. 1f. The crystal length of the prepared NaCl crystals was 150–200 μm, which strongly indicated the regulation effects of the DNA on the growth of the NaCl crystals. The NaCl crystals did not agglomerate, further indicating that DNA is an effective anticoagulant for NaCl crystal growth. The same procedure applies to DNA-2, and the optical microscopy of the prepared NaCl-2 crystals is shown in the ESI (Fig. S1†). From Fig. S1a–d,† we can also see the formation of hopper-like NaCl crystals.
The pictures shown in Fig. 2a and b are micrographs of the NaCl crystals prepared by the addition of 100 μmol L−1 DNA-1 and 1.5 mmol L−1 silver nitrate, and 100 μmol L−1 DNA-2 and 1.5 mmol L−1 silver nitrate, respectively. When we added 100 μmol L−1 DNA-1 or DNA-3 and 1.5 mmol L−1 silver nitrate to the 0.15 M NaCl solution, or added 100 μmol L−1 DNA-1 or DNA-4 and 1.5 mmol L−1 silver nitrate to the 0.15 mol L−1 NaCl solution, the NaCl crystals included not only hopper-like crystals, but also dendritic crystals (Fig. S2†). Compared with Fig. 2a and b, we can see that they all formed hopper-like NaCl crystals. However, dendritic crystals appear in Fig. S2a–d,† while not in Fig. 2a or Fig. 2b where complementary single-stranded DNA was not added. This is because DNA-1 and DNA-3 or DNA-1 and DNA-4 are hybridized following the Watson–Crick base pairing rule. Some double-stranded DNA formed to guide the crystallization of NaCl, thereby obtaining dendritic crystals aside from hopper-like crystals. In Fig. 1a and 2a, both of them used 100 μmol L−1 DNA-1 and 0.15 mol L−1 NaCl solution. The difference is that 1.5 mmol L−1 silver nitrate was added in the experiment in Fig. 2a, while not in Fig. 1a. The crystal length of the NaCl crystals in Fig. 1a is about 160 μm, while their length is about 20 μm in Fig. 2a. It can be concluded that the addition of silver nitrate can make the prepared crystals smaller.
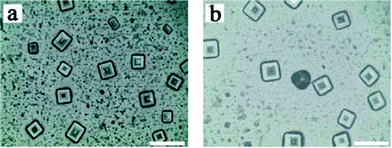 | ||
| Fig. 2 Micrographs of (a) NaCl-1 and (b) NaCl-2 prepared by 100 μmol L−1 DNA-1, 1.5 mmol L−1 silver nitrate and 100 μmol L−1 DNA-2, 1.5 mmol L−1 silver nitrate, respectively. Scale bar = 40 μm. | ||
The samples were imaged with SEM, as shown in Fig. 3. NaCl-0 crystals were prepared without adding any DNA (Fig. 3a). It was observed that the NaCl-0 particles were cubic and a large number of NaCl-0 crystals clustered together with highly heterogeneous size distribution. Crystals prepared by addition of DNA-1 and silver nitrate, as shown in Fig. 3b and c, exhibited some hopper-like surface structures, having square and rectangular morphologies. In another experiment, with the addition of DNA-2 and silver nitrate (Fig. 3d), we prepared hopper-like NaCl crystals with relatively uniform size distribution. From the high-magnification image in Fig. 3d (as well as in Fig. 3e and f), we can see that the hopper-like NaCl crystal face is formed by a regular sequence of thin growth steps. Therefore, we can conclude that additives such as DNA-1 and DNA-2 could modify the crystal morphology from a non-regular random structure to more ordered hopper-like structures with homogenized size distribution.
 | ||
| Fig. 3 SEM images of NaCl crystals (a) without DNA, (b and c) with 100 μmol L−1 DNA-1 and 1.5 mmol L−1 silver nitrate and (d–f) with 100 μmol L−1 DNA-2 and 1.5 mmol L−1 silver nitrate. | ||
The hopper-like NaCl crystals prepared in the experiment were subjected to X-ray diffraction (XRD) tests, and the product map (Fig. 4) was compared with the map of JCPDS no. 05-0628 in the database. We can see that the XRD pattern of the hopper-like NaCl crystals has no other impurity peaks, and the peaks are symmetrical, indicating that the crystal structure of the NaCl crystals is still of high quality. In particular, we can see the presence of only the {100} form in our hopper-like NaCl crystals. Combined with the existing experimental results and related literature, we deduced the DNA-mediated growth mechanism of hopper-like NaCl as follows. It has been demonstrated (through calculation) that the equilibrium shape of NaCl (in its vapor phase) can only be made by the {100}-cube form and that the adsorption of water occurring in solution cannot modify this situation.45 Moreover, it has been shown, both experimentally and theoretically, that the {111} octahedron can coexist with the cube, in specific domains of supersaturation in pure aqueous solutions. This is due to pure kinetic effects. In the experiment of Aquilano46 and co-workers, hopper-like cubes are produced along with the octahedron through the solution evaporation method. At variance with the experimental results obtained by the Aquilano group, we didn't observe the octahedron form in our {100} hopper-like crystals. Hence, we should argue that the DNA molecules have a different interaction with the cube face with respect to that with the {111} octahedron, since the cube does not have a polar face whilst the octahedron is strongly polar. Thus, the adsorption energy cannot be the same for the two faces and this surely affects the growth morphology. The experimental results show that in the supersaturated NaCl solution, DNA makes the NaCl crystals form a {100} cube. In general, there is a non-homogeneous concentration gradient in the resting solution. In our experiment, as the solution evaporates, a thin film of solution envelops the crystals on the glass. This gradient is higher in the corners, then in the edges of the crystal, and it reaches its minimum value in the middle of a flat cube face. Thus, the supersaturation values of the growth solution should be maximum in the corners and minimum in the middle of the face. Under these circumstances, the local growth rate will follow the supersaturation values. Thus, the dendrites and hoppers are generated. Moreover, Gwinn47et al. reported that Ag+ was capable of binding to four bases, adenine (A), thymine (T), cytosine (C) and guanine (G) in DNA, of which cytosine (C) was more involved in binding to Ag+ in the Cn–Agn+ –Cn mode, whereas thymine (T) was more involved in Tn–Agn+ binding. Ag+ could thus bind and even agglutinate different strands of DNA by binding to cytosine (C), whereas Ag+ bound to thymine (T) could not agglutinate the different strands of DNA. Therefore, when AgNO3 was added, the DNA tended to be aggregated. Therefore, we believe that the addition of silver ions affects the distribution of DNA, thus affecting the adsorption of DNA on NaCl {100}, resulting in a change in the size of the hopper. Starting from our observation and from the quoted theoretical considerations, we attempt to propose the following growth process. When NaCl, DNA and AgNO3 are placed together in an aqueous solution and then water evaporation is carried out at constant temperature, the solution becomes supersaturated with respect to NaCl; then, 3D NaCl nuclei form and, under the action of DNA and AgNO3 (dissociated in the solution bulk), they grow as hopper-like {100} cubes, without the presence of other forms in their growth morphology.
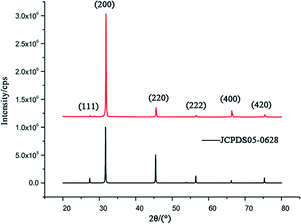 | ||
| Fig. 4 XRD patterns of hopper-like NaCl crystals prepared using 25 μl of 100 μmol L−1 DNA-1 solution and 25 μl of 1.50 mol L−1 NaCl solution (above). | ||
Conclusions
In summary, we first used DNA as an anticoagulant of NaCl crystal growth to prepare hopper-like NaCl crystals via the solvent evaporation method. In particular, the prepared hopper-like NaCl crystals are uniform. The experimental results show that the single-stranded DNA and double-stranded DNA can guide the NaCl crystals to grow into hopper-like and dendritic crystals, respectively. Besides, adding Ag+ can reduce the size of the hopper-like crystals. Through precise control over the relative concentration of NaCl and DNA, regular hopper-like NaCl crystals were generated. Therefore, this study will promote the application of DNA as an effective additive to regulate the syntheses and applications of other inorganic crystals.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by the Science Fund for Creative Research Groups of the National Natural Science Foundation of China (Grant No. 61621002) and the National Key Research and Development Program of China (2016YFC0800900, 2016YFC0800905 and 2016YFC0800905-Z03).Notes and references
- A. Sen and B. Ganguly, Angew. Chem., Int. Ed., 2012, 51, 11279–11283 CrossRef CAS PubMed
.
- R. Zheng, J. Gao, T. Yang, Y. Lan, G. Cheng, D. Wang, Z. Ren and G. Chen, Inorg. Chem., 2010, 49, 6748–6754 CrossRef CAS PubMed
.
- S. Paria, R. Ghosh Chaudhuri and N. N. Jason, New J. Chem., 2014, 38, 5943–5951 RSC
.
- Y. C. Pu, J. R. Hwu, W. C. Su, D. B. Shieh, Y. Tzeng and C. S. Yeh, J. Am. Chem. Soc., 2006, 128, 11606–11611 CrossRef CAS PubMed
.
- D. Rautaray, M. Kasture and M. Sastry, CrystEngComm, 2005, 7, 469 RSC
.
- F. Zhang, Z. Hou, K. Sheng, B. Deng and L. Xie, J. Mater. Chem., 2006, 16, 1215 RSC
.
- H. Jia, X. Bai and L. Zheng, CrystEngComm, 2011, 13, 7252 RSC
.
- X. Guo, L. Liu, W. Wang, J. Zhang, Y. Wang and S.-H. Yu, CrystEngComm, 2011, 13, 2054 RSC
.
- H. Deng, X.-M. Wang, C. Du, X.-C. Shen and F.-Z. Cui, CrystEngComm, 2012, 14, 6647 RSC
.
- F.-W. Yan, C.-Y. Guo, X.-H. Zhang and G.-Q. Yuan, CrystEngComm, 2012, 14, 1554–1560 RSC
.
- Z. Li, L. Xing, J. Xiang, X. Liang, C. Zhao, H. Sai and F. Li, RSC Adv., 2014, 4, 31210–31218 RSC
.
- Q. Liu, Y. Ma, X. Duan, Y. Zhou, X. Liu and C. Pei, CrystEngComm, 2014, 16, 11042–11049 RSC
.
- A.-X. Wang, D.-Q. Chu, L.-M. Wang, B.-G. Mao, H.-M. Sun, Z.-C. Ma, G. Wang and L.-X. Wang, CrystEngComm, 2014, 16, 5198–5205 RSC
.
- Y. Han, T. Nishimura and T. Kato, CrystEngComm, 2014, 16, 3540–3547 RSC
.
- Y.-H. Tseng, C. Chevallard, Y. Dauphin and P. Guenoun, CrystEngComm, 2014, 16, 561–569 RSC
.
- T. Sakamoto, Y. Nishimura and T. Kato, CrystEngComm, 2015, 17, 6947–6954 RSC
.
- A. Zhang, H. Xie, N. Liu, B.-L. Chen, H. Ping, Z.-Y. Fu and B.-L. Su, RSC Adv., 2016, 6, 110362–110366 RSC
.
- L.-f. Yang, D.-q. Chu, H.-l. Sun and G. Ge, New J. Chem., 2016, 40, 571–577 RSC
.
- S. Jaho, V. Sygouni, S. G. Rokidi, J. Parthenios, P. G. Koutsoukos and C. A. Paraskeva, Cryst. Growth Des., 2016, 16, 6874–6884 CAS
.
- Z. Liu, H. Pan, G. Zhu, Y. Li, J. Tao, B. Jin and R. Tang, Angew. Chem., Int. Ed., 2016, 55, 12836–12840 CrossRef CAS PubMed
.
- I. Buljan Meić, J. Kontrec, D. Domazet Jurašin, B. Njegić Džakula, L. Štajner, D. M. Lyons, M. Dutour Sikirić and D. Kralj, Cryst. Growth Des., 2017, 17, 1103–1117 Search PubMed
.
- N. Radenović, W.-V. Enckevort, P. Verwer and E. Vlieg, Surf. Sci., 2003, 523, 307–315 CrossRef
.
- A. Zafiropoulou and E. Dalas, J. Cryst. Growth, 2000, 219, 477–480 CrossRef CAS
.
- A. Ballabh, D. R. Trivedi, P. Dastidar, P. K. Ghosh, A. Pramanik and V. G. Kumar, Cryst. Growth Des., 2006, 6, 1591–1594 CAS
.
- E. R. Townsend, W. J. P. van Enckevort, J. A. M. Meijer and E. Vlieg, Cryst. Growth Des., 2015, 15, 5375–5381 CAS
.
- C. P. Fenimore and A. Thrailkill, J. Am. Chem. Soc., 1949, 71, 2714–2717 CrossRef CAS
.
- A. Giri, M. D. Choudhury, T. Dutta and S. Tarafdar, Cryst. Growth Des., 2013, 13, 341–345 CAS
.
- M. D. Choudhury, T. Dutta and S. Tarafdar, Soft Matter, 2015, 11, 6938–6947 RSC
.
- M. Goto, Y. Oaki and H. Imai, Cryst. Growth Des., 2016, 16, 4278–4284 CAS
.
- L. Chen, T. Ye, Y. J. Liu, W. Liu, G. Wu, H. Z. Chen and H. Y. Li, CrystEngComm, 2014, 16, 6901 RSC
.
- C. R. Navarro, E. Doehne and E. Sebastian, Langmuir, 2000, 16, 947–954 CrossRef
.
- P. Gu, V. S. Ramachandran, J. J. Beaudoin and E. Quinn, Adv. Cem. Based Mater., 1995, 2, 182–188 CrossRef CAS
.
- P. G. Klepetsanis and P. G. Koutsoukos, J. Cryst. Growth, 1998, 193, 156–163 CrossRef CAS
.
- C. Garcia, G. Courbin, F. Ropital and C. Fiaud, Electrochim. Acta, 2001, 46, 973–985 CrossRef CAS
.
- M. E. Tadros and I. Mayes, J. Colloid Interface Sci., 1979, 72, 245–254 CrossRef CAS
.
- J. Zhang, S. D. Zhang, Z. Y. Wang, Z. P. Zhang, S. S. Wang and S. H. Wang, Angew. Chem., Int. Ed., 2011, 50, 6044–6047 CrossRef CAS PubMed
.
- D. Kaya, V. A. Belyi and M. Muthukumar, J. Chem. Phys., 2010, 133, 114905 CrossRef CAS PubMed
.
- C. Selwitz and E. Doehne, J. Cult. Herit., 2002, 3, 205–216 CrossRef
.
- D. Aquilano, F. Otálora, L. Pastero and J. M. García-Ruiz, Prog. Cryst. Growth Charact. Mater., 2016, 62, 227–251 CrossRef CAS
.
-
M. Romé de l'Isle, Cristallographie ou description des forms propres à tous les corps du regne minéral, dans l'état de combinaison saline, pierreuse ou métalliques, Paris, 1783, p. 4 Search PubMed
.
- F. Gille and K. Spangeberg, Beiträge zur Trachtbeeinflussung des NaCl durch Harnstoff als Lösungsgenossen, Z. Kristallogr., Kristallgeom., Kristallphys., Kristallchem., 1927, 65, 204–250 CAS
.
-
M. Bienfait, R. Boistelle and R. Kern, Formes de croissance des halogénures alcalins dans un solvant polaire, in: ed. CNRS, Coll. Intern, n° 152, Adsorption et Croissance Cristalline, Paris, 1965, pp. 577–594 Search PubMed
.
- C. Jäger and H. Cölfen, CrystEngComm, 2007, 9, 1237–1244 RSC
.
- T. Kato, T. Suzuki, T. Amamiya, T. Irie, M. Komiyama and H. Yui, Supramol. Sci., 1998, 5, 411–415 CrossRef CAS
.
- M. Bruno, D. Aquilano, L. Pastero and M. Prencipe, Cryst. Growth Des., 2008, 8, 2163–2170 CAS
.
- D. Aquilano, L. Pastero, M. Bruno and M. Rubbo, J. Cryst. Growth, 2009, 311, 399–403 CrossRef CAS
.
- S. M. Swasey, L. E. Leal, O. Lopez-Acevedo, J. Pavlovich and E. G. Gwinn, Sci. Rep., 2015, 5, 10163 CrossRef CAS PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c7ce01148b |
| This journal is © The Royal Society of Chemistry 2017 |

