Alkoxido-bridged binuclear copper(II) complexes derived from aminoalcohols – useful building blocks in designing coordination polymers with a rich structural variety
Teodora
Mocanu
,
Violeta
Tudor
and
Marius
Andruh
 *
*
Inorganic Chemistry Laboratory, Faculty of Chemistry, University of Bucharest, Str. Dumbrava Rosie nr. 23, 020464-Bucharest, Romania. E-mail: marius.andruh@dnt.ro
First published on 7th June 2017
Abstract
The reactions of copper(II) salts with aminoalcohols frequently afford alkoxido-bridged dimers. Neutral (e.g. bis(4-pyridyl) derivatives) or anionic divergent ligands (e.g. polycarboxylate anions, polycyanido-metallates) are employed as spacers. In this paper, a rich variety of structures are reviewed, emphasizing the role played by the denticity of the aminoalcohols and of the linkers, as well as by the accompanying anions, in the network topology. The involvement of coordinative and non-covalent bonds (π–π stacking, argentophilic/aurophilic, π⋯Hg interactions, hydrogen bonds) in sustaining the solid-state architectures is also discussed.
Introduction
The network topologies of the coordination polymers, as well as their physical and chemical properties, are directly influenced by the metal centers and the connecting ligands. Therefore, the search for new building blocks is of continuous interest in crystal engineering, in order to reach and improve the desired properties/functions of these materials (e.g. porosity for storage and separation, catalysis, magnetism, luminescence, conductivity).1 The richness of this chemistry arises mainly from the variety of the ligands, which are organic molecules bridging the metal centers and controlling, together with the stereochemical preference of the metal ions, the directionality of the metal–ligand bonds.The rational and systematic synthesis of the coordination polymers started with the node-and-spacer approach, formulated by Robson.2 In its early stage, this strategy was developed using single metal ions, but oligonuclear complexes can also be employed as nodes. Their incorporation into extended structures occurs through the preliminary formation of a node (with a pre-established nuclearity) and subsequent reaction with a spacer, or through the serendipitous assembly of the metal ions into clusters with various nuclearities, followed by their interaction with the spacer. The spacer molecules interact with the accessible coordination sites of the metal ions by replacing coordinated solvent molecules or weakly coordinated anions. A particular case, largely illustrated by Yaghi et al.,3 consists in the formation of nodes as a result of the interaction of metal ions with spacers (e.g. the assembly of the classical binuclear paddle-wheel motif upon the interaction of carboxylate groups with copper(II) ions).
The presence of two or more metal ions within the node has important consequences: (i) original spatial arrangements of the metal–bridging ligand bonds leading to novel network topologies; (ii) redox, magnetic or electric properties arising from the metal–metal intranode and internode interactions; (iii) special versatility of the heterometallic nodes, when the metal ions differ drastically in their chemical behavior (for example, heterometallic 3d–4f nodes assembled with dissymmetric compartmental ligands).4
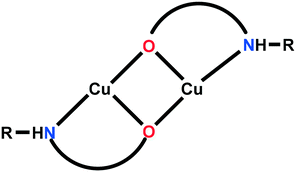
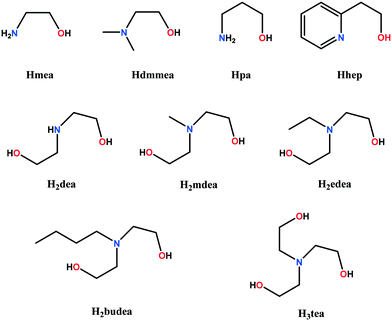
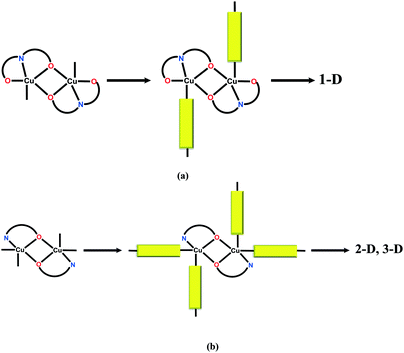
In this paper, we review the particular case of alkoxido-bridged binuclear copper(II) nodes, which are spontaneously assembled when the copper(II) salt reacts with an aminoalcohol (Scheme 1). The resulting binuclear nodes can be connected by neutral (e.g. bis(4-pyridyl) derivatives) or anionic divergent ligands (e.g. polycarboxylate anions, polycyanido-metallates). A rich library of aminoalcohols is available. Several of them, with relevance to this review, are depicted in Scheme 2. Acting simultaneously as bridging and blocking ligands while assembling the binuclear nodes, aminoalcohols control the dimensionality of the resulting networks through the number of accessible coordination sites they leave at the copper ions. For example, aminoalcohols with high denticity (H3tea, H2dea) favor the formation of 1-D coordination polymers (Scheme 3a). On the other hand, coordination polymers with higher dimensionality are obtained when aminoalcohols with lower denticity (Hmea, Hpa) are employed (Scheme 3b). Such nodes, namely, {Cu2(mea)2}2+ and {Cu2(pa)2}2+, generate 2-D and 3-D coordination polymers, provided that the anions do not coordinate to the copper ions. In this respect, copper(II) perchlorate and tetrafluoroborate are more appropriate as starting materials due to the low coordination ability of the perchlorate/tetrafluoroborate ions, although exceptions are known (see below). Conversely, when using copper(II) nitrate, the dimensionality of the resulting coordination polymers is reduced, since the nitrate ions coordinate to the metal centers as well, preventing the binding of a second linker molecule.
The binuclear nodes
The binuclear nodes can be easily isolated by performing the reaction between copper(II) salts with the aminoalcohol in the presence of monodentate or chelating ligands. Three such examples are presented in Fig. 1: [Cu(mea)(4-phpy)(ClO4)]21, [Cu(pa)(4-phpy)2]2(ClO4)22, and [Cu(pa)(dpyam)]2(ClO4)23 (4-phpy = 4-phenyl-pyridine, dpyam = dipyridylamine).5 In compound 1, one perchlorate ion forms an additional bridge between the copper ions, while the other one is a terminal ligand (the two copper ions have different coordination numbers: 5 and 6). Several binuclear complexes have been obtained using anionic terminal co-ligands: R–COO−, N3−, NCO−, NCS−, and NO2−.6 The structures of two of these are illustrated in Fig. 2: [Cu2(H2tea)2(o-MeOBz)2] 4 and [Cu2(Hbudea)2(N3)2] 5 (o-MeObz− = o-methoxybenzoate; H2budea = n-butyldiethanolamine).6b,d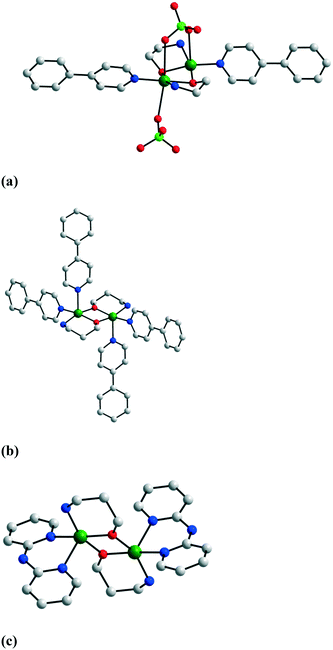 | ||
| Fig. 1 Crystal structures of binuclear complexes with neutral terminal co-ligands: (a) [Cu(mea)(4-phpy)(ClO4)]21, (b) [Cu(pa)(4-phpy)2]2(ClO4)22, and (c) [Cu(pa)(dpyam)]2(ClO4)23. Adapted from ref. 5. | ||
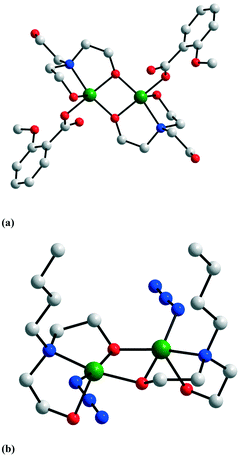 | ||
| Fig. 2 Crystal structures of binuclear complexes with anionic terminal co-ligands: (a) [Cu2(H2tea)2(o-MeOBz)2] 4 and (b) [Cu2(Hbudea)2(N3)2] 5. Adapted from ref. 6b and d. | ||
From the magnetic point of view, the alkoxido-bridged binuclear copper(II) complexes are excellent models for magneto-structural studies. Two parameters play a key role in the magnetic behavior of these compounds (nature and strength of the exchange interaction): the Cu–O–Cu angle (α) and the out-of-plane shift of the carbon atom from the bridging alkoxido group (τ angle).7 Small values of α together with large values of τ favor ferromagnetic coupling. The χMT vs. T curve for compound 1 is presented in Fig. 3. For α = 95.88 and τ = 31.9°, the exchange interaction is ferromagnetic (J = +81.9 cm−1, H = −JS1S2). Table 1 summarizes the binuclear complexes and illustrates the correlation between J and the two angles, α and τ.
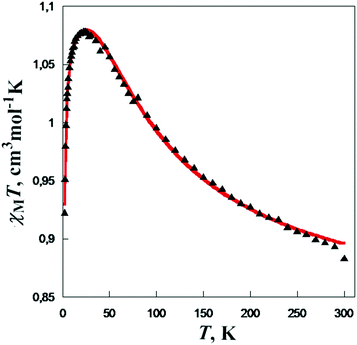 | ||
| Fig. 3 Temperature dependence of the χMT product for [Cu(mea)(4-phpy)(ClO4)]2. Adapted from ref. 5. | ||
| Complex | α (°) | τ (°) | J (cm−1) | Ref. |
|---|---|---|---|---|
| Bz− = benzoate; o-MeObz− = o-methoxybenzoate; m-MeObz− = m-methoxybenzoate; H2budea = n-butyldiethanolamine. | ||||
| [Cu2(pa)2(Bz)2] | 104.10(10) | 1.43(13) | ≥400 | 6a |
| [Cu2(Hbudea)2(N3)2] | 97.16 | 34.5 | −121 | 6d |
| [Cu2(H2tea)2(o-MeOBz)2] | 99.9 | 28 | −83.1 | 6b |
| [Cu(mea)(4-phpy)(ClO4)]2 | 95.88 | 31.9 | +81.9 | 5 |
| [Cu2(H2tea)2(m-MeOBz)2]·2H2O | 97.5 | 44 | 100.9(7) | 6b |
Several binuclear copper(II) complexes with polycarboxylate spacers are good catalysts for oxidative transformations of alkanes (cyclopentane, cyclohexane) under mild conditions,1s Some others show catecholase-like activity.6c,g
One-dimensional coordination polymers
1-D coordination polymers are assembled when the aminoalcohol has a high denticity (two or more HO groups) or when the accompanying anion blocks one of the coordination sites of the metal ion. Diethanolamine and triethanolamine are the simplest aminoalcohols which are suitable for this purpose. Let us discuss first coordination polymers assembled using neutral exo-dentate ligands: 4,4′-bipyridine (4,4′-bipy) and bis(4-pyridyl)ethylene (bpe). The structures of two representative examples are depicted in Fig. 4: [Cu2(Hdea)2(4,4′-bipy)](ClO4)26 and [Cu2(H2tea)2(bpe)](ClO4)2·(bpe)·H2O 7.8 In both compounds, the coordination of the aminoalcohol leaves, at each copper ion, only one position available for interaction with the spacer molecule. The distance between the copper ions within the nodes are 2.988 (in 6) and 2.924 Å (in 7). The distances between the nodes (11.031 and 13.402 Å in 6 and 7, respectively) are imposed by the length of the spacers. | ||
| Fig. 4 Perspective views of two 1-D coordination polymers: (a) [Cu2(Hdea)2(4,4′-bipy)](ClO4)26 and (b) [Cu2(H2tea)2(bpe)](ClO4)2·(bpe)·H2O 7. Adapted from ref. 8. | ||
As shown in Scheme 3b, monoethanolamine and propanolamine favor the formation of 2-D and 3-D coordination polymers. However, if the accompanying anion or a solvent molecule blocks one of the coordination sites at the copper ions, the dimensionality is reduced to 1. Fig. 5 illustrates the structures of 1-D coordination polymers found in [Cu2(pa)2(py2Hg)(ClO4)2]·0.5(py2Hg)·H2O 8 (ref. 9) and [Cu2(mea)2(bpbenz)(NO3)](NO3)·2.75CH3OH 9,10 where py2Hg = bis(4-pyridyl)mercury and bpbenz = p-bis(4-pyridyl)benzene. In compound 8, one perchlorate ion is coordinated to each copper ion, while in 9 the nitrate ligand forms an additional bridge between the copper ions. An interesting coordination polymer was found in [Cu4(pa)4(bpp)4(ClO4)2](ClO4)2·2H2O 10 (Fig. 6), where bpp = bis(4-pyridyl)propane.11 Although the chemical composition and the employment of propanolamine suggest the formation of a 2-D coordination polymer, the crystallographic investigation revealed chains with two different alkoxido-bridged centrosymmetric binuclear nodes: in one type of node the fifth coordination site at each copper ion is occupied by a perchlorate ligand (the distance between the copper ions is 2.979 Å), while in the second type the fifth coordination site is occupied by a terminal bpp ligand (the distance between the copper ions is 3.058 Å).
 | ||
| Fig. 5 Perspective views of the coordination polymers [Cu2(pa)2(py2Hg)(ClO4)2]n and [Cu2(mea)2(bpbenz)(NO3)]nn+ in crystals 8 (a) and 9 (b). Adapted from ref. 9 and 10. | ||
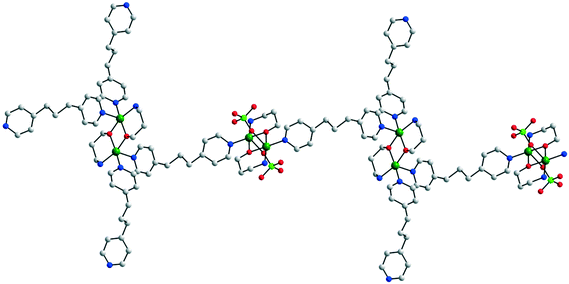 | ||
| Fig. 6 View of the 1-D coordination polymer in [Cu4(pa)4(bpp)4(ClO4)2](ClO4)2·2H2O 10. Adapted from ref. 11. | ||
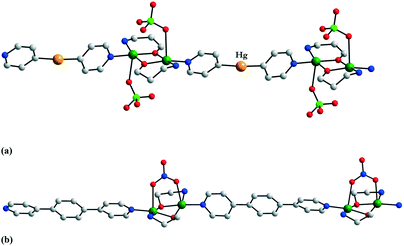
The uncoordinated spacer molecules influence the solid-state architecture of the crystals, being involved in various supramolecular interactions: the nitrogen atom from the bis(4-pyridyl) derivatives can act as a hydrogen bond acceptor, and the mercury atom from py2Hg can be involved in π⋯Hg interactions. For the particular case of compound 8, the mercury–centroid distance is 3.49 Å. Two relevant packing diagrams for crystals 7 and 8 are displayed in Fig. 7.
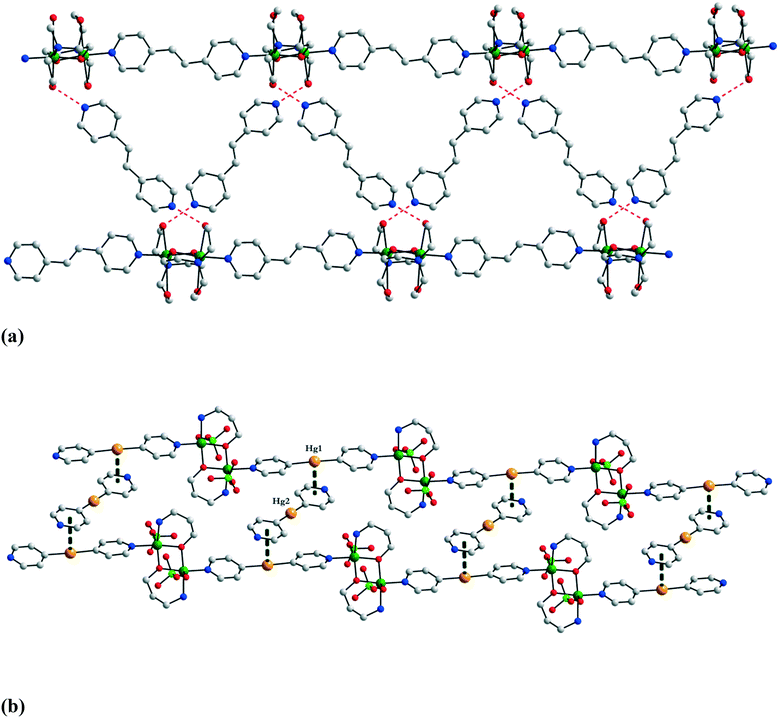 | ||
| Fig. 7 Packing diagrams for crystals 7 (a) and 8 (b). The mercury–centroid distance in 8 is 3.49 Å. Adapted from ref. 8 and 9. | ||
A 1-D coordination polymer with an original topology was assembled using the exo-tridentate ligand tris(3-pyridyl)benzene (tpyb): [Cu2(mea)2(tpyb)(ONO2)](NO3)·CH3OH 11 (Fig. 8a).10 The tpyb ligand is coordinated to three copper ions belonging to three different nodes, resulting in a double chain with alternating small and large metallacycles. Since the pyridyl moieties of the tpyb ligand are not coplanar with the central benzene ring, the framework shows a tube-like structure (Fig. 8b).
 | ||
| Fig. 8 (a) View of the structure of [Cu2(mea)2(tpyb)(ONO2)](NO3)·CH3OH 11; (b) alternative view of 11 showing the tube-like structure. Adapted from ref. 10. | ||
Anionic spacers, such as polycarboxylate ions (terephthalate, isophthalate, 2,6-naphalenedicarboxylate, trimesate), are also employed. Several 1-D coordination polymers with dicarboxylate linkers have been described by Seppälä et al.6a and by Pombeiro, Kirillov et al.,6d,12 one of them being illustrated in Fig. 9: [Cu2(ap)2(ipa)(dmso)2]·dmso 12 (H2ipa = isophthalic acid).6a If the deprotonation of the polycarboxylic acid is attained with sodium hydroxide, the sodium ions can be incorporated into the polymeric framework; the reaction between copper(II) nitrate, triethanolamine, and isophthalic acid in the presence of NaOH leads to two compounds: [Cu2(H2tea)2(ipa)]·H2O 13a (main product) and [Cu2(H2tea)2{Na(H2O)3}(ipa)](NO3)·0.5H2O 13b (minor product).12b Compound 13a is made from {Cu2(H2tea)2}2+ nodes and ipa2− spacers. In compound 13b parallel [Cu2(H2tea)2(ipa)]n chains are interconnected by dimeric {Na2(μ-H2O)2(H2O)4}2+ moieties, each sodium ion being coordinated, apart from the aqua ligands, by an oxygen atom arising from a carboxylate group and by one OH group from the H2tea− ligand. The intranode exchange interaction between the copper ions in 13a was found to be ferromagnetic (J = +60 cm−1), the α and τ angles being 96.67 (average value) and 31.9°, respectively. A similar development in two directions, through the intervening sodium ions, was found in the heterometallic 2-D coordination polymer [Cu2(H2tea)2(pma)Na2(H2O)4]·10H2O (pma4− = pyromellitate).13
 | ||
| Fig. 9 Structure of [Cu2(ap)2(ipa)(dmso)2] 12, a coordination polymer constructed from {Cu2(pa)2} nodes and isophthalato spacers (adapted from ref. 6a). | ||
Other 1-D coordination polymers have been assembled from binuclear nodes generated by 2-(2-hydroxyethyl)pyridine (Hhep), a ligand that shows a coordination behavior similar to that described before for mea− and pa−.14 These networks result from the linking of the nodes by the anions NO3−, CO32−, and C6H5–OCH2COO−: [Cu2(hep)2(μ-NO3)2] 14, [Cu2(hep)2(H2O)2(μ-CO3)]·2H2O 15, and [Cu2(hep)2(μ-PhOAc)2] 16 (Scheme 4).
Two-dimensional coordination polymers
When the binuclear nodes are assembled using aminoalcohols with only two donor groups, 2-D coordination polymers are readily obtained. Numerous grid-like sheets have been obtained by employing rigid bis(4-pyridyl) derivatives: pyrazine (pyr), 4,4′-bipyridine, 1,2-bis(4-pyridyl)ethylene (bpe), p-bis(4-pyridyl)benzene (bpbenz), bis(4-pyridyl)acetylene (bpac), and 9,10-bis(4-pyridyl)anthracene (bpanth). The measurements of the grid cell are determined by the length of the spacers. Several representative examples are illustrated in Fig. 10: [Cu2(mea)2(pyr)2](CF3SO3)217,11 [Cu2(ap)2(4,4′-bipy)2](ClO4)3·(4,4′-bipy)·(H2ap)·(H2O) 18,8 [Cu2(mea)2(bpe)2](ClO4)219,15 [Cu2(mea)2(bpbenz)2](CF3SO3)2·0.5(bpbenz)·3CH3OH 20,10 and [Cu2(pa)2(bpac)2](ClO4)221.10 Analysis of the chemical composition of these compounds raises the question about the way the sheets pack in the crystal. There are two families of crystals: those containing only the coordination polymers and the accompanying anions, and those incorporating, in addition, organic guest molecules (most frequently uncoordinated spacer molecules). In the first case, interpenetration of the 2-D sheets is observed, resulting in an interlocked 3-D structure. Fig. 11 shows the inclined interpenetration observed in crystal 21, which is described as a 2D inclined catenation p–p (parallel–parallel) of (4,4) layers with a Doc of 2/2:16 every mesh of every sheet contains parts of two others passing through it. The interpenetration is favored by long spacer ligands. In the second case, the 2-D networks stack parallel to one another, generating channels which are filled by the guest molecules. Fig. 12 shows the superposition of the layers in crystal 18. Each channel is filled by an interesting H-bonded supramolecular polymer constructed from uncoordinated 4,4′-bipy and water molecules, H2pa+ (monoprotonated propanolamine species) and perchlorate ions.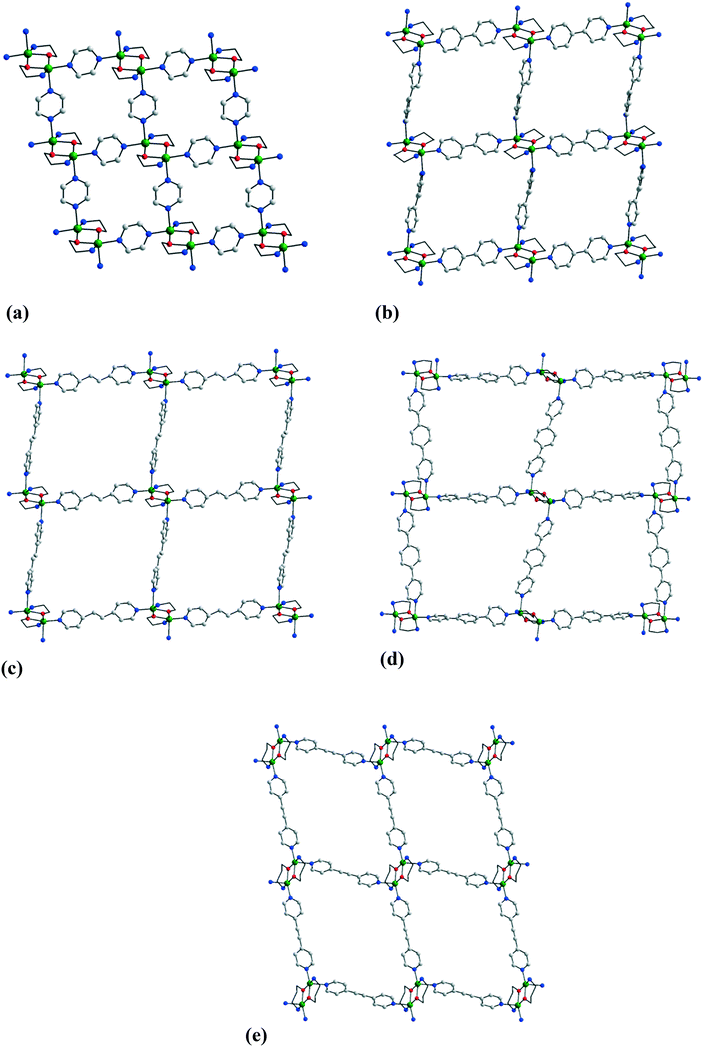 | ||
| Fig. 10 Grid-like coordination polymers constructed from rigid linear spacers: (a) [Cu2(mea)2(pyr)2](CF3SO3)217, (b) [Cu2(ap)2(4,4′-bipy)2](ClO4)3(4,4′-bipy)·(H2ap)·(H2O) 18, (c) [Cu2(mea)2(bpe)2](ClO4)219, (d) [Cu2(mea)2(bpbenz)2](CF3SO3)2·0.5(bpbenz)·3CH3OH 20, and (e) [Cu2(pa)2(bpac)2](ClO4)221. Adapted from ref. 8, 10, 11 and 15. | ||
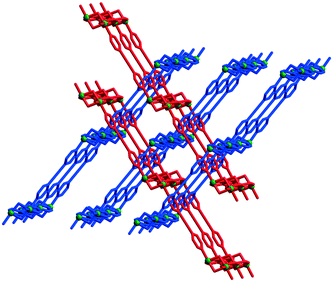 | ||
| Fig. 11 Inclined interpenetration of the grid-like layers in [Cu2(pa)2(bpac)2](ClO4)221. Adapted from ref. 10. | ||
 | ||
| Fig. 12 Packing diagram for crystal 18, showing the formation of channels. Adapted from ref. 8. | ||
What happens when the exo-dentate ligand is angular and flexible? To answer this question, we employed bis(4-pyridyl)disulfide (bpds) to connect {Cu2(mea)2}2+ or {Cu2(pa)2}2+ nodes. Let us describe here the structure of [Cu2(mea)2(bpds)2](CF3SO3)2·2CH3OH 22.10 It is built from centrosymmetric binuclear nodes and angular bpds spacers with a C–S–S–C torsion angle of 87.49° (Fig. 13a). The structure can be alternatively viewed as comprising copper centers connected by the bpds ligands in helicoidal chains, which are then interconnected by the alkoxido bridges to form layers. A view of the layer along the crystallographic b axis reveals the formation of chiral channels which are filled by methanol molecules (each layer contains Cu–bpds helices of opposite chiralities – Fig. 13b).
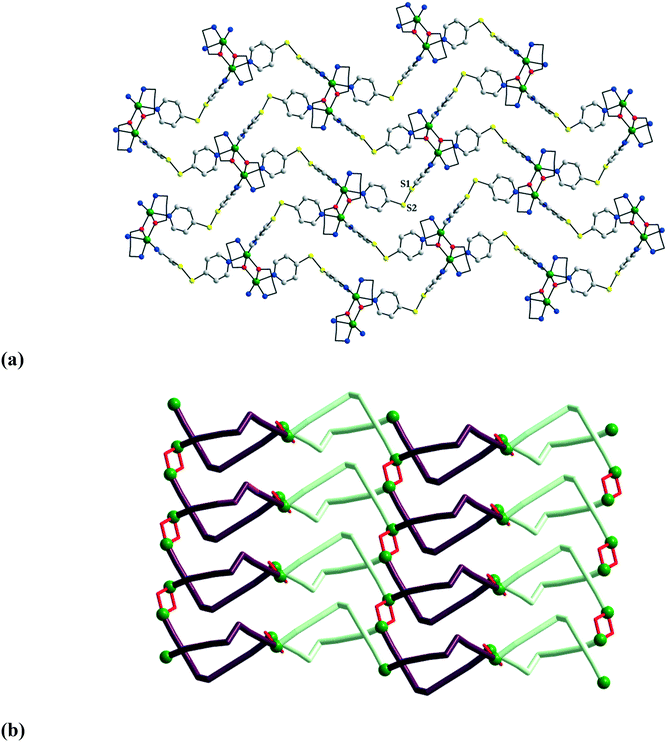 | ||
| Fig. 13 View of the 2-D coordination polymer [Cu2(mea)2(bpds)2]nn+ in 22 (a); side view showing the helicoidal chains (b). Adapted from ref. 15. | ||
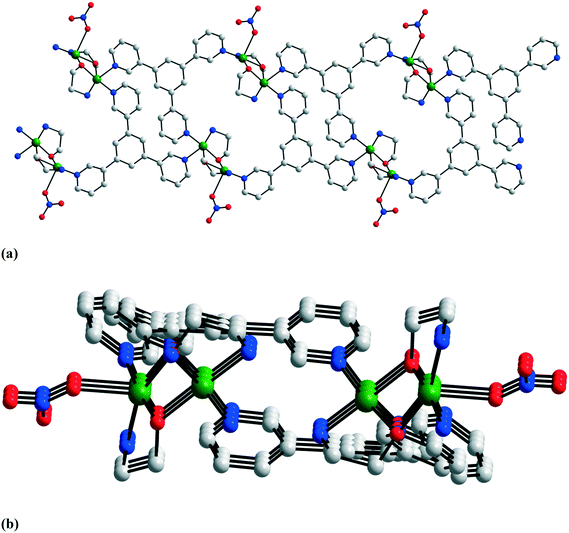
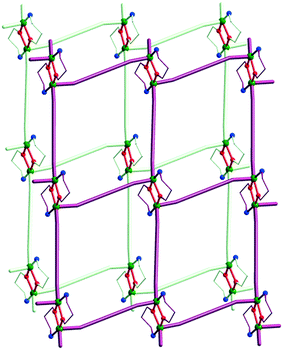
An interesting case concerning the architecture of the crystal is represented by the isostructural coordination polymers constructed from {Cu2(pa)2}2+ nodes and linear anionic spacers [M(CN)2]− (M = Ag, Au): [Cu2(pa)2M(CN)2] 23a and b.17 The overall topology of the layers (Fig. 14) is similar to the one observed with neutral linear spacers (compounds 17–21). The packing of the layers in the crystal is, however, different. Both compounds consist of neutral polymeric networks, without anions or neutral molecules to be hosted. The arrangement of the layers is driven by aurophilic (argentophilic) interactions, the gold (or silver) atoms from one layer being disposed exactly above the gold (or silver atoms) from another layer (Fig. 15). The shortest contacts between the silver and gold atoms in 23a and 23b are Ag⋯Ag = 3.015 and Au⋯Au = 3.069 Å.
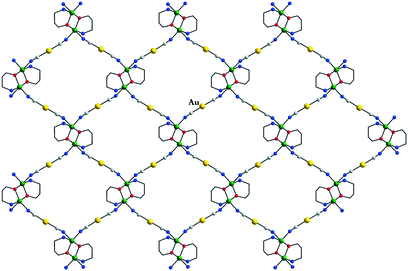 | ||
| Fig. 14 Grid-like layers in [Cu2(pa)2Au(CN)2] 23b. Adapted from ref. 17. | ||
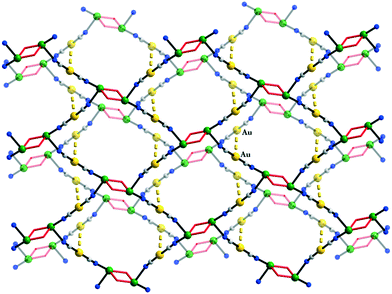 | ||
| Fig. 15 Packing diagram for [Cu2(pa)2Au(CN)2] 23b showing the aurophilic interactions between the layers. Adapted from ref. 17. | ||
The NO2− ion, with a versatile coordination behavior, can act as a bridging ligand. The one pot reaction between copper(II) perchlorate, propanolamine, and NaNO2 leads to [Cu2(ap)2(NO2)2] 24, a 2-D coordination polymer with a brick-wall topology (Fig. 16).6a,18 The intranode exchange interaction between the copper ions is moderately antiferromagnetic (J = −302 cm−1; α = 99.54(6); τ = 32.26(12)).6a
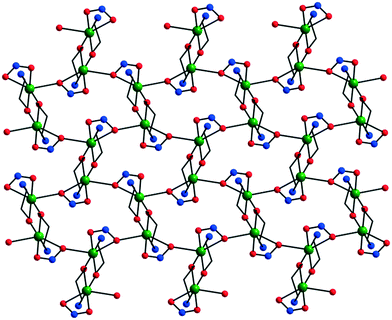 | ||
| Fig. 16 Crystal structure of the coordination polymer [Cu2(ap)2(NO2)2] 24. Adapted from ref. 18. | ||
Recently, Kirillov et al.19 described neutral bidimensional networks which are generated by the anion of the pyromellitic acid, pma4−: [{Cu2(dmmea)2(H2O)}(pma)]·4H2O 25 and [Cu2(Hedea)2(pma)]·4H2O 26 (Hdmmea = N,N-dimethyl-ethanolamine; Hdedea = N-ethyl-diethanolamine). Their topologies are similar, with the structure of 25 being depicted in Fig. 17. The carboxylate groups from the pma4− linkers have different coordination behaviors: two of them coordinate to the copper ion through only one oxygen atom, while the two others coordinate with both oxygen atoms (chelating).
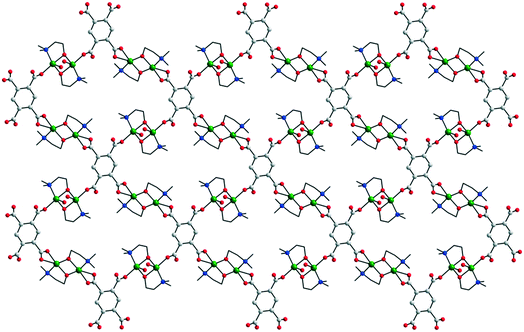 | ||
| Fig. 17 View of the 2-D network in [{Cu2(dmmea)2(H2O)}(pma)]·4H2O 25. Adapted from ref. 19. | ||
Three-dimensional coordination polymers
The number of 3-D coordination polymers is limited to only a few examples. The first 3-D network built from binuclear alkoxido-bridged nodes and a linear spacer was obtained by reacting Cu(BF4)2 with propanolamine and bis(4-pyridyl)mercury: [Cu2(pa)2(py2Hg)2](BF4)227.9 Two py2Hg linkers are coordinated to each copper(II) ion, resulting in a cadmium sulphate topology (Fig. 18a). Four interpenetrating nets are observed in the packing diagram (Fig. 18b).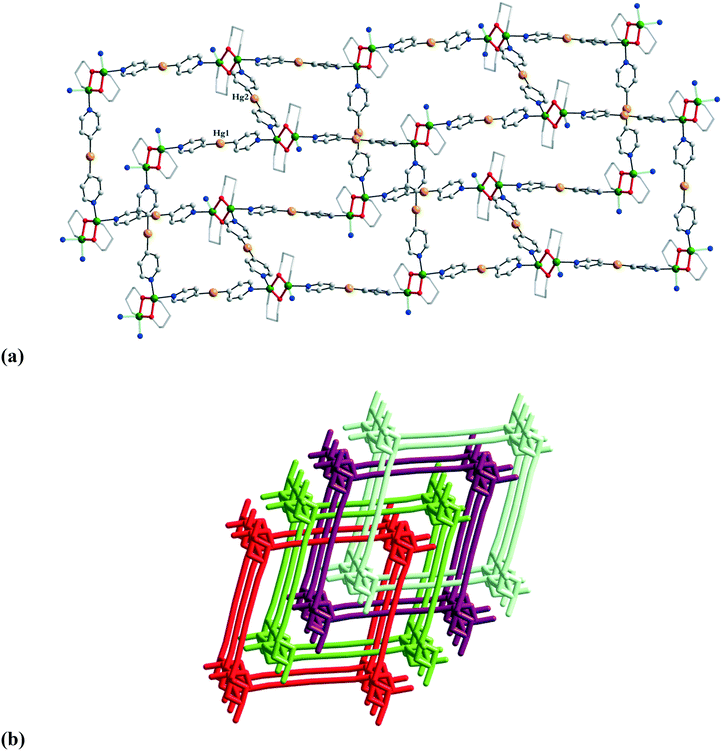 | ||
| Fig. 18 Three-dimensional network with cadmium sulphate topology in [Cu2(pa)2(py2Hg)2](BF4)227 (a); packing diagram showing the four-fold interpenetration in crystal 27 (b). Adapted from ref. 9. | ||
In principle, divergent ligands with more than two potentially bridging groups favor the formation of 3-D networks. Such linkers can be homoleptic polycyanide complexes. Indeed, the self-assembly process involving [Cu2(pa)2]2+ and [Ni(CN)4]2− ions leads to [Cu2(pa)2{Ni(CN)4}] 28, featuring a 3-D coordination architecture.17
The extended structure of 28 is built up of two different centrosymmetric {Cu2(pa)2} nodes, Cu1 and Cu2, and of [Ni(CN)4]2− ions with two different connecting modes (Ni1 and Ni2). Taking into account the strength of the Cu–NC interactions, the crystal structure can be described as follows: the basal positions of the copper ions from Cu1 nodes are connected by two trans cyanide groups from Ni1 blocks, while the copper ions from the Cu2 nodes are connected through their basal positions by two cis cyanide groups from Ni2 anions, resulting in 2-D layers (Fig. 19a). The other two trans CN groups from Ni1 bridge the apical positions of the copper ions from neighbouring Cu2 units within the same chain. The structure is developed into the third dimension by connecting the apical positions of the copper ions from adjacent Cu1 nodes through the remaining cis CN groups from the Ni2 anions (Fig. 19b).
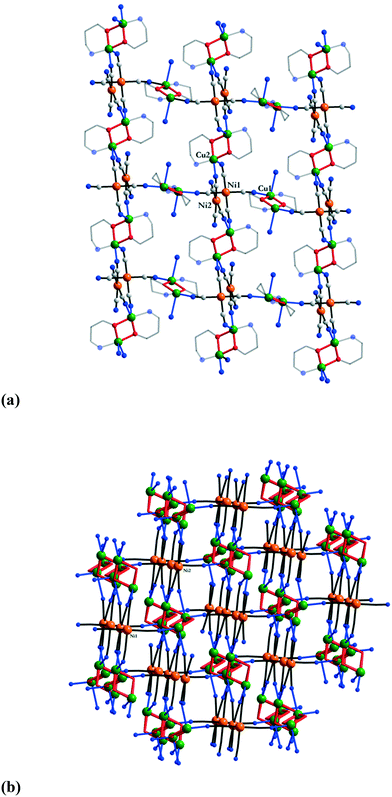 | ||
| Fig. 19 Views of the 3-D network in [Cu2(pa)2{Ni(CN)4}] 28: (a) connectivity modes; (b) 3-D network. Adapted from ref. 17. | ||
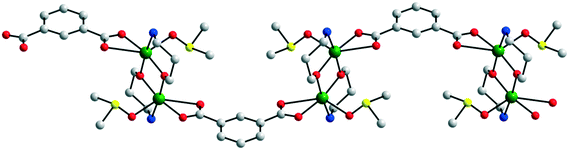
Two other heterobimetallic 3-D coordination polymers were obtained using [Fe(CN)6]4− linkers: [{Cu2(H2tea)2}3{Fe(CN)6}](NO3)2·6H2O 29 and [{Cu2(Hmdea)2}3{Fe(CN)6}](NO3)2·7H2O 30 (Hmdea = N-methyl-diethanolamine).20 In compound 29, each [Fe(CN)6]4− ion interacts, through its cyanide groups with one copper ion from six binuclear nodes, {Cu2(H2tea)2}, resulting in a regular octahedral net structure (Fig. 20), with channels hosting the nitrate ions and water molecules. The structure of 30 is similar but more distorted. After dehydration, both compounds adsorb water or methanol reversibly. The magnetic properties of compounds 29 and 30 arise from the intranode exchange interaction between the copper ions. In compound 29 the interaction was found to be antiferromagnetic, while in compound 30 the copper ions are ferromagnetically coupled (Fig. 21). The difference originates from the values of the Cu–O–Cu angles within the two compounds: α = 101.33° in 29 (J = −199(1) cm−1), and α = 90.5° (average) in 30 (J = +153(1) cm−1).20
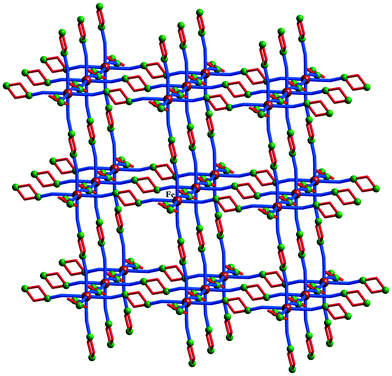 | ||
| Fig. 20 View of the 3-D network in [{Cu2(Hmdea)2}3{Fe(CN)6}](NO3)2·7H2O 30. Adapted from ref. 20. | ||
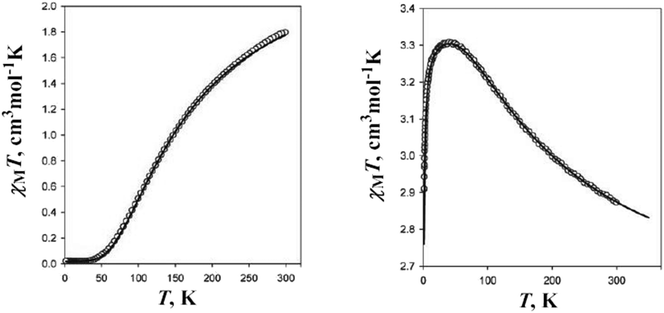 | ||
| Fig. 21 χ M T vs. T curves for compounds 29 and 30.20 | ||
Conclusions and perspectives
Aminoalcohols can form mononuclear complexes, but their ability to aggregate clusters with two or more metal ions, bridged by alkoxido groups, is much more attractive. The chemistry generated by aminoalcohols and copper(II) ions is quite rich. In this paper, we focused on the binuclear alkoxido-bridged species which are robust tectons for constructing coordination polymers. The network topologies are directed by the denticity of the aminoalcohols, the number and the spatial arrangements of the donor atoms from the spacer molecules, as well as by the accompanying anions. The structural and electronic peculiarities of the spacer molecules also influence the solid-state architecture through various non-covalent interactions.The diversity of the coordination polymers already reported in the literature and of those to be synthesized arises from the large choice of aminoalcohols and, in particular, from the very rich library of exo-dentate ligands that can be employed (poly-pyridyl derivatives, polycarboxylates, inorganic anions, cyanido-metalloligands). This library can be enlarged with divergent ligands featuring different donor atoms (for example, pyridyl-carboxylates, or other heterocyclic compounds carrying carboxylate groups). Moreover, this synthetic approach based on binuclear nodes can be extended towards other 3d metal ions, as we have shown recently.21
The aminoalcohols generate not only binuclear copper(II) species. Actually, their coordination chemistry is much broader and of high interest. Numerous oligonuclear complexes with more than 2 copper(II) ions are known and can serve as nodes in crystal engineering.17,22
We learn, from this chemistry too, that, in order to obtain porous crystals, a good strategy consists of avoiding the presence of uncoordinated anions, which occupy the channels or cavities. Consequently, cationic binuclear nodes must be connected by anionic spacers. Another way is to design neutral nodes and then connect them by neutral spacers. Indeed, we obtained a neutral binuclear alkoxido-bridged complex by reacting copper(II) perchlorate with mandelic acid in the presence of ammonia. The hydroxyl group from mandelic acid forms the alkoxido bridge. The neutral binuclear nodes have been subsequently connected by hexamethylenetetramine spacer molecules, resulting in a robust porous 3-D MOF.23
Although several heterobimetallic coordination polymers have been obtained using diamagnetic cyanido-metalloligands, systems assembled from alkoxido-bridged {CuII2} nodes and another paramagnetic metal ion have not been reported so far. These compounds can be particularly interesting from the magnetic point of view, if the intranode copper–copper interaction is ferromagnetic. The binuclear complexes themselves represent an excellent platform for magnetostructural correlations.
References
-
(a) R. A. Agarwal and N. K. Gupta, Coord. Chem. Rev., 2017, 332, 100 CrossRef CAS
; (b) N. S. Bobbitt, M. L. Mendonca, A. J. Howarth, T. Islamoglu, J. T. Hupp, O. K. Farha and R. Q. Snurr, Chem. Soc. Rev., 2017, 46, 3357 RSC
; (c) T. R. Cook, Y.-R. Zheng and P. J. Stang, Chem. Rev., 2013, 113, 734 CrossRef CAS PubMed
; (d) E. Coronado and G. M. Espallargas, Chem. Soc. Rev., 2013, 42, 1525 RSC
; (e) F.-X. Coudert, Chem. Mater., 2015, 27, 1905 CrossRef CAS
; (f) J. Duan, W. Jin and S. Kitagawa, Coord. Chem. Rev., 2017, 332, 48 CrossRef CAS
; (g) M. L. Foo, R. Matsuda and S. Kitagawa, Chem. Mater., 2014, 26, 310 CrossRef CAS
; (h) G. Givaja, P. Amo-Ochoa, C. J. Gómez-García and F. Zamora, Chem. Soc. Rev., 2012, 41, 115 RSC
; (i) Y. Hasegawa and T. Nakanishi, RSC Adv., 2015, 5, 338 RSC
; (j) C. He, D. Liu and W. Lin, Chem. Rev., 2015, 115, 11079 CrossRef CAS PubMed
; (k) J. Heine and K. Müller-Buschbaum, Chem. Soc. Rev., 2013, 42, 9232 RSC
; (l) Z. R. Herm, E. D. Bloch and J. R. Long, Chem. Mater., 2014, 26, 323 CrossRef CAS
; (m) C. Janiak, Dalton Trans., 2003, 2781 RSC
; (n) S. Kitagawa, R. Kitaura and S. Noro, Angew. Chem., Int. Ed., 2004, 43, 2334 CrossRef CAS PubMed
; (o) W. L. Leong and J. J. Vittal, Chem. Rev., 2011, 111, 688 CrossRef CAS PubMed
; (p) Z. Ma and B. Moulton, Coord. Chem. Rev., 2011, 255, 1623 CrossRef CAS
; (q) A. Mahmood, W. Guo, H. Tabassum and R. Zou, Adv. Energy Mater., 2016, 1600423 CrossRef
; (r) L. R. Mingabudinova, V. V. Vinogradov, V. A. Milichko, E. Hey-Hawkins and A. V. Vinogradov, Chem. Soc. Rev., 2016, 45, 5408 RSC
; (s) A. M. Kirillov, M. V. Kirillova and A. J. L. Pombeiro, Coord. Chem. Rev., 2012, 256, 2741 CrossRef CAS
.
-
(a) R. W. Gable, B. F. Hoskins and R. Robson, J. Chem. Soc., Chem. Commun., 1990, 1677 RSC
; (b) B. F. Hoskins and R. Robson, J. Am. Chem. Soc., 1990, 112, 1546 CrossRef CAS
; (c) R. Robson, Dalton Trans., 2008, 5113 RSC
.
- See, for example:
(a) M. O'Keeffe and O. M. Yaghi, Chem. Rev., 2012, 112, 675 CrossRef PubMed
; (b) D. J. Tranchemontagne, Z. Ni, M. O'Keeffe and O. M. Yaghi, Angew. Chem., Int. Ed., 2008, 47, 5136 CrossRef CAS PubMed
; (c) J. Jiang, H. Furukawa, Y.-B. Zhang and O. M. Yaghi, J. Am. Chem. Soc., 2016, 138, 10244 CrossRef CAS PubMed
; (d) A. Schoedel, M. Li, D. Li, M. O'Keeffe and O. M. Yaghi, Chem. Rev., 2016, 116, 12466 CrossRef CAS PubMed
.
-
(a) M. Andruh, Pure Appl. Chem., 2005, 77, 1685 CrossRef CAS
; (b) M. Andruh, Chem. Commun., 2007, 2565 RSC
; (c) M. Andruh, Chem. Commun., 2011, 47, 3015 RSC
; (d) M. Andruh, D. G. Branzea, R. Gheorghe and A. M. Madalan, CrystEngComm, 2009, 11, 2571 RSC
; (e) R. Gheorghe, P. Cucos, M. Andruh, J.-P. Costes, B. Donnadieu and S. Shova, Chem.-Eur. J., 2006, 12, 187 CrossRef CAS PubMed
; (f) M.-G. Alexandru, D. Visinescu, M. Andruh, N. Marino, D. Armentano, J. Cano, F. Lloret and M. Julve, Chem.-Eur. J., 2015, 21, 5429 CrossRef CAS PubMed
; (g) M. Andruh, Chimia, 2013, 67, 383 CrossRef CAS PubMed
.
- V. Tudor, T. Mocanu, F. Tuna, A. M. Madalan, C. Maxim, S. Shova and M. Andruh, J. Mol. Struct., 2013, 1046, 164 CrossRef CAS
.
-
(a) P. Seppälä, E. Colacio, A. J. Mota and R. Sillanpää, Inorg. Chim. Acta, 2010, 363, 755 CrossRef
; (b) R. P. Sharma, A. Saini, P. Venugopalan, V. Ferretti, F. Spizzo, C. Angeli and C. J. Calzado, New J. Chem., 2014, 38, 574 RSC
; (c) Y.-H. Chung, H.-H. Wei, Y. H. Liu, G.-H. Lee and Y. Wang, J. Chem. Soc., Dalton Trans., 1997, 2825 RSC
; (d) K. R. Gruenewald, A. M. Kirillov, M. Haukka, J. Sanchiz and A. J. L. Pombeiro, Dalton Trans., 2009, 2109 RSC
; (e) V. T. Yilmaz, Y. Topcu, F. Yilmaz and C. Thoene, Polyhedron, 2001, 20, 3209 CrossRef CAS
; (f) P. Figiel, A. M. Kirillov, M. F. C. Guedes da Silva, J. Lasri and A. J. L. Pombeiro, Dalton Trans., 2010, 39, 9879 RSC
; (g) C.-H. Kao, H.-H. Wei, G.-H. Lee, Y. Wang and C.-J. Lee, J. Inorg. Biochem., 2001, 84, 171 CrossRef CAS PubMed
; (h) M. Mikuriya, M. Yamamoto and W. Mori, Bull. Chem. Soc. Jpn., 1994, 67, 1384 Search PubMed
.
- E. Ruiz, P. Alemany, S. Alvarez and J. Cano, J. Am. Chem. Soc., 1997, 119, 1297 CrossRef CAS
.
- V. Tudor, G. Marin, V. Kravtsov, Yu. A. Simonov, J. Lipkowski, M. Brezeanu and M. Andruh, Inorg. Chim. Acta, 2003, 353, 35 CrossRef CAS
.
- T. Mocanu, C. I. Raţ, C. Maxim, S. Shova, V. Tudor, C. Silvestru and M. Andruh, CrystEngComm, 2015, 17, 5474 RSC
.
- G. Marin, M. Andruh, A. M. Madalan, A. J. Blake, C. Wilson, N. R. Champness and M. Schröder, Cryst. Growth Des., 2008, 8, 964 CAS
.
- G. Marin, V. Kravtsov, Yu. A. Simonov, V. Tudor, J. Lipkowski and M. Andruh, J. Mol. Struct., 2006, 796, 123 CrossRef CAS
.
-
(a) S. S. P. Dias, V. André, J. Kłak, M. T. Duarte and A. M. Kirillov, Cryst. Growth Des., 2014, 14, 3398 CrossRef CAS
; (b) A. M. Kirillov, Y. Y. Karabach, M. Haukka, M. F. C. Guedes da Silva, J. Sanchiz, M. N. Kopylovich and A. J. L. Pombeiro, Inorg. Chem., 2008, 47, 162 CrossRef CAS PubMed
.
- Y. Y. Karabach, A. M. Kirillov, M. F. C. Guedes da Silva, M. N. Kopylovich and A. J. L. Pombeiro, Cryst. Growth Des., 2006, 6, 2200 CAS
.
- S. M. Mobin, A. K. Srivastava, P. Mathur and G. K. Lahiri, RSC Adv., 2011, 1, 893 RSC
.
- G. Marin, V. Tudor, V. Ch. Kravtsov, M. Schmidtmann, Yu. A. Simonov, A. Müller and M. Andruh, Cryst. Growth Des., 2005, 5, 279 CAS
.
- L. Carlucci, G. Ciani and D. M. Proserpio, Coord. Chem. Rev., 2003, 246, 247 CrossRef CAS
.
- C. Paraschiv, M. Andruh, S. Ferlay, M. W. Hosseini, N. Kyritsakas, J.-M. Planeix and N. Stanica, Dalton Trans., 2005, 1195 RSC
.
- L. J. Farrugia, D. S. Middlemiss, R. Sillanpää and P. Seppälä, J. Phys. Chem. A, 2008, 112, 9050 CrossRef CAS PubMed
.
- T. A. Fernandes, C. I. M. Santos, V. André, J. Kłak, M. V. Kirillova and A. M. Kirillov, Inorg. Chem., 2016, 55, 125 CrossRef CAS PubMed
.
- Y. Y. Karabach, M. F. C. Guedes da Silva, M. N. Kopylovich, B. Gil-Hernández, J. Sanchiz, A. M. Kirillov and A. J. L. Pombeiro, Inorg. Chem., 2010, 49, 11096 CrossRef CAS PubMed
.
- C. Paraschiv, A. Cucos, S. Shova, A. M. Madalan, C. Maxim, D. Visinescu, B. Cojocaru, V. I. Parvulescu and M. Andruh, Cryst. Growth & Des., 2015, 15, 799 CAS
.
- See, for example:
(a) P. Seppälä, E. Colacio, A. J. Mota and R. Sillanpää, Dalton Trans., 2012, 41, 2648 RSC
; (b) P. Seppälä, E. Colacio, A. J. Mota and R. Sillanpää, Inorg. Chem., 2013, 52, 11096 CrossRef PubMed
; (c) Z. Chen, L. Liu, Y. Wang, H. Zou, Z. Zhang and F. Liang, Dalton Trans., 2014, 43, 8154 RSC
; (d) S. S. P. Dias, M. V. Kirillova, V. André, J. Kłak and A. M. Kirillov, Inorg. Chem., 2015, 54, 5204 CrossRef CAS PubMed
; (e) V. Tudor, V. Ch. Kravtsov, M. Luve, F. Lloret, Yu. A. Simonov, B. B. Averkiev and M. Andruh, Inorg. Chim. Acta, 2005, 358, 2066 CrossRef CAS
; (f) J. K. Maclaren, J. Sanchiz, P. Gili and C. Janiak, New J. Chem., 2012, 36, 1596 RSC
.
- E. Ilyes, M. Florea, A. M. Madalan, I. Haiduc, V. I. Parvulescu and M. Andruh, Inorg. Chem., 2012, 51, 7954 CrossRef CAS PubMed
.
| This journal is © The Royal Society of Chemistry 2017 |